The experimentally determined density of O 2 at 140. bar and 298 K is 192 g L
Question:
Figure 7.8
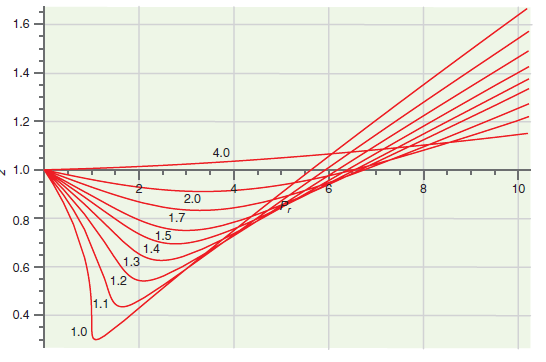
Transcribed Image Text:
1.6 1.4 1.2 4.0 v 1.0 8. 10 2.0 0.8 1.7 1.5 1.4 1.3 0.6 1.2 1.1 0.4 1.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Because P V 140 Bar5043 bar 278 and T V 298 ...View the full answer

Answered By

Rohail Amjad
Experienced Finance Guru have a full grip on various sectors, i.e Media, Insurance, Automobile, Rice and other Financial Services.
Have also served in Business Development Department as a Data Anlayst
4.70+
32+ Reviews
83+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
At 725 K and 280. bar, the experimentally determined density of N 2 is 4.13 mol L 1 . Compare this with values calculated from the ideal and RedlichKwong equations of state. Use a numerical equation...
-
Calculate the density of O 2 (g) at 480. K and 280. bar using the ideal gas and the van der Waals equations of state. Use a numerical equation solver to solve the van der Waals equation for V m or...
-
For the 2-kg connecting rod shown, it has been experimentally determined that the mass moments of inertia of the rod with respect to the center-line axes of the bearings AA and BB are, respectively,...
-
Welch Company prepares monthly financial statements. Below are listed some selected accounts and their balances in the September 30 trial balance before any adjustments have been made for the month...
-
What are the advantages and disadvantages of the single-step income statement?
-
You are doing x-ray diffraction on a crystal that has a cubic structure, using \(0.500-\mathrm{nm} x\) rays. If the lattice spacing is \(d=6.70 \times 10^{-10} \mathrm{~m}\), what are the two...
-
A governmental auditor is performing an audit of the city of Pawtucket, Rhode Island. The auditor's objective is to determine whether the city's financial statements are presented fairly in all...
-
Ritz Productss materials manager, Tej Dhakar, must determine whether to make or buy a new semiconductor for the wrist TV that the firm is about to produce. One million units are expected to be...
-
1- Workpapers (year of adquisicion, fair value/book value differentials, intercompanu balances) Pik Corporation acquired 80 percent of Sel Corporation's common stock on January 1, 2011, for $210,000...
-
1. How comparable are the two different methods? In what ways are they similar? In what ways are they different? 2. What are the positive and negative aspects of each approach that Shocker should...
-
A business has assets of $1,000,000 and liabilities of $600,000. What is the amount of owners equity in this business? a. $400,000 b. $1,400,000 c. $600,000 d. $1,600,000
-
A 1.75 mole sample of Ar undergoes an isothermal reversible expansion from an initial volume of 2.00 L to a final volume of 85.00 L at 310. K. Calculate the work done in this process using the ideal...
-
Calculate the marginal propensity to consume. At what level of disposable income will saving be zero? If expected future income increases, in which direction will the consumption function change? The...
-
analyze the attached Seagate case. In your analysis, you should address the following questions: 1) Why is Seagate undertaking this transaction? Is it necessary to divest the Veritas shares in a...
-
Please answer the following questions: a) Impairment tests are required for particular investments at the year-end. Indicate which investment does not require impairment test and explain why? b)...
-
Respond to Brittany's discussion in a friendly way: Brittany's discussion, The three items from the course I am taking with me are the utilization of Cash Budgets, the power of Compound Interest, and...
-
Budgeted manufacturing overhead costs $4,300,000 Budgeted machine-hours 172,000 Actual manufacturing overhead costs $4,140,000 Actual machine-hours 166,000 pop-up content ends. 1. Calculate the...
-
ABC Inc. reported $75 million in revenue for last year. On average, over the same year, the company had $25 million worth of inventory in their warehouses. Assume that units in inventory are valued...
-
Evaluate the integral. dx
-
Heineken N.V., a global brewer based in the Netherlands, reports the following balance sheet accounts for the year ended December 31, 2016 (euros in millions). Prepare the balance sheet for this...
-
Evaluate the triple integral in cylindrical polar coordinates: 3.00 4.00 I = zp cos? (4) d dp dz.
-
Find the volume of a right circular cylinder of radius a = 4.00 with a paraboloid of revolution scooped out of the top of it such that the top surface is given by z = 10.00 + 1.00 2 and the bottom...
-
Find the volume of a solid with vertical walls such that its base is a square in the xy plane defined by 0 x 2.00 and 0 x 2.00 and its top is defined by the plane z = 20.00 + x + y.
-
Mercury Company has only one inventory pool. On December 31, 2024, Mercury adopted the dollar-value LIFO inventory method. The inventory on that date using the dollar-value LIFO method was $201,000....
-
Arizona Corporation acquired the business Data Systems for $315,000 cash and assumed all liabilities at the date of purchase. Data's books showed tangible assets of $330,000, liabilities of $18,000,...
-
Define and describe the concepts of IMC (Integrated Marketing Communications) and RTM (real time marketing). How does this relate to the discussion in the textbook of time compression and online vs....

Study smarter with the SolutionInn App


