The following kinetic data (v 0 is the initial rate) were obtained for the reaction 2 ICl(g)+H
Question:
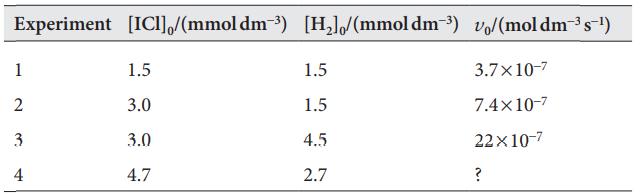
The following kinetic data (v0 is the initial rate) were obtained for the reaction 2 ICl(g)+H2(g)→I2(g)+2 HCl(g):
Transcribed Image Text:
Experiment [IC]/(mmol dm-³) [H₂]/(mmoldm-³) v/(moldm³s-¹) 1 2 3 4 1.5 3.0 3.0 4.7 1.5 1.5 4.5 2.7 3.7x10-7 7.4×10-7 22x10-7 ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
The maximum velocity is given by VmaxkcatE where Vmax is the maximum velocity kcat i...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
The following data were obtained for the reaction 2 ICl ( g ) + H 2 ( g ) I 2 ( g ) + 2 HCl ( g ) Experiment [ICl] 0 (M) [H 2 ] 0 (M) Initial Rate (M/s) 1 0.0015 0.0015 3.7 x 10 -7 2 0.0030 0.0015...
-
The following data were obtained for the reaction 2ClO2(aq) + 2OH2(aq) ClO3-(aq) + ClO2-(aq) + H2O(l) where a. Determine the rate law and the value of the rate constant. b. What would be the initial...
-
The following data were obtained for the reaction A B, catalyzed by the enzyme Aase. The reaction volume was 1 mL and the stock concentration of A was 5.0 mM. Seven separate reactions were examined,...
-
Solve Problem using augmented matrix methods. Use a graphing calculator to perform the row operations 5.7x1 - 8.55x2 = -35.91 4.5x1 +5.73x2 = 76.17
-
On a day off from work, an employee visited the store at which she was employed for the purpose of signing a sympathy card for a coworker. While talking with coworkers at the back of the store, a...
-
The following information was drawn from the accounting records of Connor Manufacturing Company. During the accounting period, Connor paid $16,000 to purchase raw materials, $15,000 for direct labor,...
-
How many degrees of freedom are there? Exercises 49 refer to the following data: Electric motors are assembled on four different production lines. Random samples of motors are taken from each line...
-
Famas Llamas has a weighted average cost of capital of 8.9 percent. The companys cost of equity is 12 percent and its cost of debt is 7.9 percent. The tax rate is 35 percent. What is Famas target...
-
Perform a first derivative test on the function f(x) = 2x3 +9x2-24x+3; [-4,5] a. Locate the critical points of the given function. b. Use the First Derivative Test to locate the local maximum and...
-
The management at Ohio National Bank does not want its customers to wait in line for service for too long. The manager of a branch of this bank estimated that the customers currently have to wait an...
-
Consider a polymer formed by a chain process. By how much does the kinetic chain length change if the concentration of initiator increases by a factor of 3.6 and the concentration of monomer...
-
Distinguish between a pre-equilibrium approximation and a steady-state approximation. Why might they lead to different conclusions?
-
Adamson Company manufactures four lines of garden tools. As a result of an activity analysis, the accounting department has identified eight activity cost pools. Each of the product lines is produced...
-
Bernard co. has 9% coupon bonds on the market that have 13 years left to maturity. The bonds will make annual payments. If the YTM on these bonds is 6%, what is the current bond price (in $ dollars)?...
-
ABC common stock is expected to have extraordinary growth in earnings and dividends of 19% per year for 2 years after which the growth rate will settle into a constant 4% if the discount rate is 13%...
-
A rope joins two toboggans. The first toboggan has a 65 kg crate on it and the second has a similar 25 kg crate. A 75 N force is applied to the first toboggan resulting in both moving forward....
-
Over the past 3 years an investment returned 0.16, -0.12, and 0.05. What is the variance of returns?
-
A uniform 3,984 N piece of medical apparatus that is 3.5m long is suspended horizontally by two vertical wires at its ends. A small but dense 383 N weight is placed on the apparatus 2.0m from one...
-
Frigid Motors Inc. assembles and sells snowmobile engines. The company began operations on July 1, 2016, and operated at 100% of capacity during the first month. The following data summarize the...
-
You are maintaining a subsidiary ledger account for Police-Training Expenditures for 2013. The following columns are used: Inventory purchases are initially recorded as expenditures. Record the...
-
Just as for the finite depth box, wave functions for which E-V 0 is small (3s) penetrate further into the barrier than wave functions for which E-V 0 is large (1s). The 2s wave function is...
-
What are the units of the H atom total energy eigenfunctions? Why is a 0 3/2 R(r) graphed in Figure 20.6 rather than R(r)?
-
Why is the radial probability function rather than * (r) (r) r 2 sin dr d d the best measure of the probability of finding the electron at a distance r from the nucleus?
-
Balance, September 1 Checks BANK STATEMENT Deposits Other Balance $ 1,940 September 7 NSF check $ 90 1,850 September 11 $ 2,910 4,760 September 12 #101 $ 770 3,990 September 17 #102 1,640 2,350...
-
A large hospital required nurses to work 80 hours over two-week (14 day) pay periods. In addition, nurses were also considered to be "on-call" for an additional 20 hours during the pay period. While...
-
Describe the history of the HIPAA, explaining why it is essential. Consider the following questions in your response: Why is understanding the history of the HIPAA law essential in decision making...

Study smarter with the SolutionInn App


