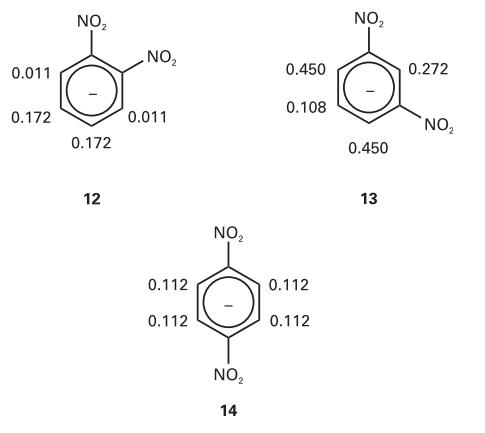
The hyperfine coupling constants observed in the radical anions (12), (13), and (14) are shown (in millitesla,
Question:
The hyperfine coupling constants observed in the radical anions (12), (13), and (14) are shown (in millitesla, mT). Use the value for the benzene radical anion to map the probability of finding the unpaired electron in the π orbital on each C atom.
Transcribed Image Text:
0.011 0.172 NO ₂ 0.172 12 NO₂ 0.011 0.112 0.112 NO ₂ NO ₂ 14 0.450 0.108 0.112 0.112 NO₂ 0.450 13 0.272 NO₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
Solution For radical anions hyperfine coupling constants are give...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The hyperfine coupling constant in CH3 is 2.3 mT. Use the information in Table 15.3 to predict the splitting between the hyperfine lines of the spectrum of CD what are the overall widths of the...
-
The hyperfine interaction in a hydrogen atom between the magnetic dipole moment of the proton and the spin magnetic dipole moment of the electron splits the ground level into two levels separated by...
-
The relative orbital levels for the hydrogen atom can be represented as Draw the relative orbital energy levels for atoms with more than one electron, and explain your answer. Also explain how the...
-
Prove that, the area of the traverse is equal to the algebraic sum of the products of the total latitude of each point and algebraic sum of the departures of the lines meeting at that point.
-
Discussion Questions: 1. After returning from the training session at Hamburger University, a McDonalds store owner selected a random sample of 362 drive-thru customers and carefully measured the...
-
Consider the following frequency table of observations on the random variable X. Values 0 1 2 3 4 Observed Frequency 24 30 31 11 4 (a) Based on these 100 observations, is a Poisson distribution with...
-
Predict the value of y when x1 = 20, x2 = 20, and x3 = 30. Exercises 1115 refer to the following data set: x1 *2 x3 69.8 7.9 37.3 62.4 32.3 9.3 20.2 40.7 66.9 13.3 30.5 48.7 87.5 27.4 38.8 35.8 93.5...
-
Preston Media Corporation had the following income statement and balance sheet for 2018: Requirements 1. Compute the acquisition of plant assets for Preston Media Corporation during 2018. The...
-
Determine the total cost pool for BRANCH 2 after allocating the costs from the support departments using the reciprocal method Determine the total cost pool for BRANCH 1 after allocating the costs...
-
Figure P2.22 shows two pendulums suspended from frictionless pivots and connected at their midpoints by a spring [1]. Assume that each pendulum can be represented by a mass M at the end of a mass...
-
Sketch the appearance of the 1 H-NMR spectrum of acetaldehyde (ethanal) using J = 2.90 Hz and the data in Exercise 15.9a in a spectrometer operating at (a) 250 MHz, (b) 500 MHz. Data in Exercise...
-
The chemical shift of the CH 3 protons in acetaldehyde (ethanal) is = 2.20 and that of the CHO proton is 9.80. What is the difference in local magnetic field between the two regions of the molecule...
-
Prove that (9.147) implies (x l), (x m) = 0 for all l m. (4(x), 4(x m)) = 4(x) 4(x m) dx = 0 for all m = 0. (9.147)
-
ABC Company has the following mutually exclusive projects. If the company's payback period is 2 years, which of these projects should be chosen? Project A Year 0 -19,500 1 11,500 2 8,750 3 2,500...
-
8.Home Depot offers an installment loan at 4.39% (add-on rate) if you buy a refrigerator. If the refrigerator at Home Depot costs $2377 and you want to pay for the refrigerator for 4 years, what is...
-
A Bank currently offers traditional banking services, from which they generate an average return of 8% while bearing a risk () of 2%. They are planning to allocate 45% of their activity to...
-
5.Consider a Blazer that is advertised at a monthly payment of $1714 for 5 year with 8.88%. What is the APR (to the nearest tenth of a percent)? 7.You want to purchase a computer that has a price of...
-
1. Please post your thoughts on the Inner Work Life Reading - https://hbr.org/2007/05/inner-work-life-understanding-the-subtext-of-business-performance 2. Please post your thoughts on the following...
-
Commercially available antiserum samples containing antibodies for antigens A, B, and D are used to determine the type of a particular patient's blood. The antiserum is mixed with the sample of...
-
In the simple quantity theory of money, what will lead to an increase in aggregate demand? In monetarism, what will lead to an increase in aggregate demand?
-
The dissociation vapour pressure ofNH4Cl at 427C is 608 kPa but at 459C it has risen to 1115 kPa. Calculate (a) The equilibrium constant, (b) The standard reaction Gibbs energy, (c) The standard...
-
Estimate the temperature at which CuS045H,O undergoes dehydration.
-
For PbI2(s) = 0Pb+(aq) + 2 r(aq), K = 1.4 X 10-8 at 25C and the standard Gibbs energy of formation ofPbI2(s) is -173.64 k] mol ". Calculate the standard Gibbs energy of formation of PbI2 (aq).
-
On March 1, 2017 Green Company purchased a new piece of equipment for 210,000 cash. On March 1 Green recorded the equipment purchase with a Debit to the Equipment account and a Credit to the Cash...
-
The price per share of your all-equity firm is $40, and there are 2M shares outstanding. Suppose that your firm issues $30M worth of debt. The debt has a face value of $30M, a coupon rate of 5...
-
Provide a detailed explanation explaining the importance of organizational politics in the workplace. I am providing two references that you can use in your response. Please provide the pros and cons...

Study smarter with the SolutionInn App


