The mean activity coefficients for aqueous solutions of NaCl at 25C are given below. Confirm that they
Question:
The mean activity coefficients for aqueous solutions of NaCl at 25°C are given below. Confirm that they support the Debye–Hückel limiting law and that an improved fit is obtained with the extended law.
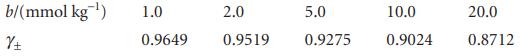
Transcribed Image Text:
b/(mmol kg ¹) 土 1.0 0.9649 2.0 0.9519 5.0 0.9275 10.0 0.9024 20.0 0.8712
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Solution Here we know k A 12ln 12k A 05885 ln 1 04021 ln 2 Since the DebyeHckel law is v...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The mean activity coefficients for aqueous solutions of NaCl at 25 C are given opposite. Confirm that they support the DebyeHckel limiting law and that an improved fit is obtained with the Davies...
-
The volume of an aqueous solution of NaCl at 25 C was measured at a series of molalities b, and it was found that the volume fitted the expression v=1003+16.62x+1.77x 3/2 +0.12x 2 where v=V/cm 3 , V...
-
The mean activity coefficients of KCI in three dilute aqueous solutions at 25C are 0.927 (at 5.0 mmol kg-I), 0.902 (at 10.0 mmol kg-I), and 0.816 (at 50.0 mmol kg"). Estimate the value of B in the...
-
Zolezzi Incorporated is preparing its cash budget for March. The budgeted beginning cash balance is $29,000, Budgeted cash receipts total $100,000 and budgeted cash disbursements total $91,000. The...
-
Why does a dealer offer to trade only a fixed amount at the bid and ask prices?
-
In a certain process 2.15 X 105 J of heat is liberated by a system, and at the same time the system contracts under a constant external pressure of 9.50 X 105 Pa. The internal energy of the system is...
-
The General Social Survey reported that in a sample of 68 men aged 1825, the mean number of hours of television watched per day was 2.76 with a standard deviation of 2.21. In a sample of 72 women...
-
In this chapter, you learned about the importance of testing. Design a generic test plan that describes the testing for an imaginary system.
-
Courtney has decided that she wishes to travel to Malaysia for 4 months when she finishes university. Courtney has estimated that her trip expenses will be $1500.00 per month of travel, and she will...
-
Three orchards supply crates of oranges to four retailers. The daily demand amounts at the four retailers are 150, 150, 400, and 100 crates, respectively. Supplies at the three orchards are dictated...
-
The enthalpy of fusion of anthracene is 28.8 kJ mol 1 and its melting point is 217C. Calculate its ideal solubility in benzene at 25C.
-
The osmotic pressure of solutions of polystyrene in toluene were measured at 25C and the pressure was expressed in terms of the height of the solvent of density 1.004 g cm 3 : Calculate the molar...
-
What methods are commonly used for roof drainage?
-
A cargo container is 25ft long, 10 ft tall and 12ft wide. Find its volume in cubic yards. Round to the nearest hundredth
-
discuss and analyse the key cultural and environmental issues which could influence a selected company's approach to digital marketing in the selected country. One should address the impact on...
-
Intrinsic motivation is when we are motivated by internal factors and the rewards are internal like joy or satisfaction. Extrinsic motivation is when we are motivated by external factors like a grade...
-
Using the base case, calculate the composite effects of a 5 percent increase in Sales, a 2 percent decrease in Gross Margin, and a 5 percent increase is SG&A as percent of Sales. Colossal Chemical...
-
An athlete throws a shot with weight m kg. At the beginning of the throwing motion, the shot is held h m above the ground. The athlete raises the shot to a height of h m before releasing it. The shot...
-
Why is this important?
-
The outer loop controls the number of students. Note that the inner loop of this program is always executed exactly three times, once for each day of the long weekend. Modify the code so that the...
-
As a variation of the preceding problem explore the consequences of increasing the energy separation of the B and C orbitals (use S=0 for this stage of the calculation). Are you justified in ignoring...
-
Write down the secular determinants for (i) anthracene (1), (ii) phenanthrene (2) within the Hckel approximation and using the C2p orbitals as the basis set. 1 Anthracene 2 Phenanthrene
-
What is the physical significance of the Coulomb and resonance integrals?
-
Curl Up & Dye, Inc. began May with $200 of supplies. It purchased $830 of supplies on account during May. It paid $440 of the amount it owed for its supplies in May. At May 31, it only had $200 of...
-
Where do traditional Waterfall approaches work effectively? Detailed explanations and site examples.
-
The accounting records for a restaurant indicate that food sales were $18,000, food used was $5,800, and employee meals at cost were $50. What is the cost of sales?

Study smarter with the SolutionInn App


