The reaction mechanism involves an intermediate A. Deduce the rate law for the reaction. AA+A (fast) A+B
Question:
The reaction mechanism
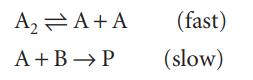
involves an intermediate A. Deduce the rate law for the reaction.
Transcribed Image Text:
A₂A+A (fast) A+B → P (slow)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The mechanism of the following transformation involves a carbocation intermediate that rearranges in a way that we have not yet seen. Rather than occurring via a methyl shift or a hydride shift, a...
-
The rate law for the reaction 2NOBr(g) 2NO(g) + Br2(g) at some temperature is a. If the half- life for this reaction is 2.00 s when [NOBr]0 = 0.900 M, calculate the value of k for this reaction. b....
-
The rate law for the reaction BrO3-(aq) + 3SO32-(aq) Br2(aq) + 3SO4 2-(aq) is Rate = k[BrO3-][SO32-][H+] The first step in a proposed mechanism is The second step is rate determining. Write a...
-
All else equal, a large sell off in the equity markets (decline in the S&P 500 and the Dow Jones Industrial Average) would be the most likely result from the reporting of a much higher than expected...
-
Students who live on campus and purchase a meal plan are randomly assigned to one of three dining halls: the Commons, Northeast, and Frazier. What is the probability that the next student to purchase...
-
What is the average current drawn by a 1.0-hp 120-V motor? (1 hp = 746 W.)
-
Are conventional financial accounting and external ecological accounting complements to each other or substitutes for each other? Discuss.
-
A gas-turbine power plant operates on the simple Brayton cycle between the pressure limits of 100 and 1200 kPa. The working fluid is air, which enters the compressor at 30°C at a rate of 150...
-
Here are the relevant estimates for the budget preparation of Beta Corporation for the fourth quarter of 2023: October November December Credit sales 1 403 380 1 631 850 1 205 930 Credit purchases...
-
CAT Company is considering two independent business projects; which are constructing a luxury condominium (project A) and building a plaza avenue (similar to J-Avenue) (project B). The financial...
-
Show that t 1/2 1/[A] n1 for a reaction that is nth-order in A.
-
A reaction 2 A P has a second-order rate law with k = 3.50 10 4 dm 3 mol 1 s 1 . Calculate the time required for the concentration of A to change from 0.260 mol dm 3 to 0.011 mol dm 3 .
-
You have been asked to write a brief report on how TQM can be adopted by your organization to improve data quality. Produce a list of reasons why TQM should and should not be adopted, and recommend,...
-
What is the 529 college savings plans? And how does the 529 affect Taxation of qualified tuition plan contributions and distributions.
-
Discuss why your orange farm business is likely a perfectly competitive firm. Describe a scenario where you would want to stay in business despite operating at a loss. [Hint: this has to do with the...
-
This advertisement by "Reporters without Borders" (a non-profit group that supports free media globally) refers to the manipulation of media by Vladimir Putin's government. The message says,...
-
The table lists the state income tax rates for a single person earning $50,000 per year for several states in 2019. State Income Tax (%) Arizona 3.34 California 8.00 Hawaii 8.25 Georgia 5.75 New...
-
The following are probable salvage value estimates for an asset: $4,500 with a probability of 33%, and $7,800 with a probability of 67%. Compute the "expected" salvage value of the asset.
-
a. Is a private placement more likely to be used for issues of seasoned stock or seasoned bonds by an industrial company? b. Is a rights issue more likely to be used for an initial public offering or...
-
During the month, services performed for customers on account amounted to $7,500 and collections from customers in payment of their accounts totaled $6,000. At the end of the month, the Accounts...
-
The rate of consumption of B in the reaction A + 3 B 7 C + 2 D is 1.0 mol dm3 S-1. State the reaction rate, and the rates of formation or consumption of A, C, and D.
-
The rate law for the reaction in Exercise 22.1b was found to be v = k[A][BF, What are the units of k? Express the rate law in terms of the rates of formation and consumption of (a) A, (b) C.
-
The rate law for the reaction in Exercise 22.2b was reported as d[C]/dt = k[A] [B][C]-l Express the rate law in terms of the reaction rate; what are the units for k in each case?
-
Build a GUI conference registration program: StuPre | Conference Registration Event # of Participants Select Event 1 1 Event 2 2 Event 3 0 Event 4 1 Event 5 0 Enter your email: Register
-
1. Write a method that takes a string as input and prints true if the String length is greater than 6 characters. The method should print false otherwise. 2. Write a method that takes a String s and...
-
The analysis algorithm is known as follows: function Analysis(n:integer) ->integer {Initial State: n >= 0 Final State: Return calculation n(n+1)/2} Dictionary i: integer res: real Algorithm Res

Study smarter with the SolutionInn App


