The reaction rate as a function of initial reactant pressures was investigated for the reaction 2NO(g) +
Question:
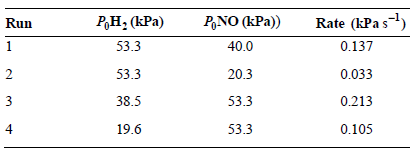
What is the rate law expression for this reaction?
Transcribed Image Text:
Rate (kPa s) 0.137 P,NO (kPa)) Р,Н: (КPa) Run 53.3 40.0 2 53.3 20.3 0.033 38.5 53.3 0.213 0.105 53.3 19.6 4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
The rate law expression is Rate kP H2 P NO We first find the order of the reaction U...View the full answer

Answered By

Usman Nasir
I did Master of Commerce in year 2009 and completed ACCA (Association of Chartered Certified Accountants) in year 2013. I have 10 years of practical experience inclusive of teaching and industry. Currently i am working in a multinational company as finance manager and serving as part time teacher in a university. I have been doing tutoring via many sites. I am very strong at solving numerical / theoretical scenario-based questions.
4.60+
16+ Reviews
28+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The reaction H2SeO3(aq) + 6I2(aq) + 4H+(aq) Se(s) + 2I3-(aq) + 3H2O(l) was studied at 0oC, and the following data were obtained: [Io Initial Rate (mol/L) (mol I1 s-1) (molVL (moVL.) 1.0 x10 2.0 x...
-
For the reaction I (aq) + OCl (aq) OI (aq) + Cl (aq) occurring in aqueous solution, the following mechanism has been proposed: a. Derive the rate law expression for this reaction based on this...
-
The reaction 2NO(g) + O2(g) 2NO2(g) was studied, and the following data were obtained, where Rate = d[O2]/dt What would be the initial rate for an experiment where [NO]0 = 6.21 Ã 1018...
-
what is resourced based view in public administration and why is it important?
-
In Exercises 1-6, match the polynomial function with its graph. [The graphs are labeled (a), (b), (c), (d), (e), and (f).] a. b. c. d. e. f. 1. f (x) = 2x2 5x 2. f (x) = 2x3 3x + 1 3. f (x) = 1/4x4 +...
-
a. Sometimes, our discussion of marginal cost and marginal revenue unintentionally hides the real issue: the entrepreneurs quest to maximize total profits. Here is information on a firm: Demand: P =...
-
With reference to Exercise 11.9, (a) find a 95% confidence interval for the mean current density when the strain is \(x=0.50\); (b) find 95% limits of prediction for the current density when a new...
-
Mississippi River Shipyards is considering replacing an 8-year-old riveting machine with a new one that will increase earnings before depreciation from $27,000 to $54,000 per year. The new machine...
-
The following T-accounts record the operations of Vaughn Co.: Direct Materials Beginning Balance 29,000 ? 244,000 Ending Balance 18,000 Beginning Balance Work in Process 37,000 Direct Material ?...
-
A fireboat draws seawater (SG =1.025) from a submerged pipe and discharges it through a nozzle, as in Fig. P3.137. The total head loss is 6.5 ft. If the pump efficiency is 75 percent, what horsepower...
-
What is the overall order of the reaction corresponding to the following rate constants? a. k = 1.63 10 4 M 1 s 1 b. k = 1.63 10 4 M 2 s 1 c. k = 1.63 10 4 M 1 2 s 1
-
One loss mechanism for ozone in the atmosphere is the reaction with the HO2 radical: HO 2 · (g) + O 3 (g) OH · (g) + 2O 2 (g) Using the following information, determine the rate law...
-
What type of contract is riskiest for the contractor? Why?
-
Explain the difference between absolute and gage pressures.
-
Describe the nature and purpose of a personal safety plan.
-
Describe the nature and potential significance of an at-risk adoption.
-
Explain when it may be appropriate to seek a guardianship in a family violence case.
-
In early 2019, policymakers at the Federal Reserve forecast that real GDP during 2019 would increase at the same rate as potential GDP and that the inflation rate for the year would be about 1.8...
-
Wind erosion is more likely in an arid environment than in a more humid environment. What other factors might impede wind erosion?
-
The activities listed in lines 2125 serve primarily as examples of A) Underappreciated dangers B) Intolerable risks C) Medical priorities D) Policy failures
-
In each of the following cases, determine if the base is sufficiently strong to deprotonate the terminal alkyne. That is, determine whether the equilibrium favors formation of the alkynide ion. (a)...
-
The pK a of CH 3 NH 2 is 40, while the pK a of HCN is 9. (a) Explain this difference in acidity. (b) Can the cyanide anion (the conjugate base of HCN) be used as a base to deprotonate a terminal...
-
For a gas that obeys the equation of state V m = Rt/P + B(T) derive the result dB(T) = B(T) T d
-
Use the formula for the general term (the nth term) of an arithmetic sequence to find the sixth term of the sequence with the given first term and common difference. a = 10; d=6 86
-
The increasing of the insurance premium has raised a great concern of the public. As the general manager of General Insurance Agency Inc., you would like to use the balanced scorecard to improve the...
-
Your Client Mr. Smith has booked the appointment with your associate and is exploring a variety of Investment and retirement planning options. He wants to better understand the offerings available to...

Study smarter with the SolutionInn App


