The relative permittivities of methanol (m.p. 95C) corrected for density variation are given below. What molecular information
Question:
The relative permittivities of methanol (m.p. −95°C) corrected for density variation are given below. What molecular information can be deduced from these values? Take ρ = 0.791 g cm−3 at 20°C.
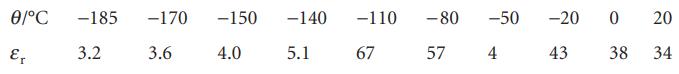
Transcribed Image Text:
Ꮎ/°C -185 Ꮛ 3.2 -170 -150 -140 3.6 4.0 5.1 -110 -80 67 57 -50 -20 4 43 0 38 20 34
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
The two strongest absorption bands for methane occur at 3967 and 4...View the full answer

Answered By

Enock Oduor
I am a chemist by profession, i coach high school students with their homework, i also do more research during my free time, i attend educational and science fair seminars where i meet students and do some projects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Journalize the following transactions: a. Issued credit memo No. 2 to Rose, $50. b. Cash sales, $210 c. Received cheque from Tran Co., $90, less 3% discount. d. Bought merchandise on account from Mel...
-
The variation in the density of water, p , with temperature, T, in the range 20°C ¤ T ¤ 50°C, is given in the following table. Use these data to determine an empirical equation...
-
For following molecular model, where C atoms are black and H atoms are light blue: a. Write the molecular formula. b. Write the condensed structural formula. c. Write the IUPAC name.
-
The following information is available for Marigold's Hot Dogs: Actual production Budgeted production Standard direct labor hours Actual direct labor hours Standard variable overhead rate Actual...
-
Suppose a recent random sample of employees nationwide that have a 401(k) retirement plan found that 18% of them had borrowed against it in the last year. A random sample of 100 employees from a...
-
A thin uniform copper rod of length l and mass m rotates uniformly with an angular velocity in a horizontal plane about a vertical axis passing through one of its ends. Determine the tension in the...
-
The accounting records for ADR, Inc., reflected the following amounts at the end of August 2010: Prepare ADRs multi-step income statement for the fiscal year ended August 31,2010. Cash...... $3,500...
-
For the customers in Exercise, In exercise At a major credit card bank, the percentages of people who historically apply for the Silver, Gold, and Platinum cards are 60%, 30%, and 10%, respectively....
-
As detailed above, many banks expressed concern about including unrealized gains and losses on AFS securities in the calculation of regulatory capital. The Federal Reserve Board noted: "Generally,...
-
The Willow Furniture Company produces tables. The fixed monthly cost of production is $8,000, and the variable cost per table is $65. The tables sell for $180 apiece. a. For a monthly volume of 300...
-
Suppose an H 2 O molecule ( = 1.85 D) approaches an anion. What is the favourable orientation of the molecule? Calculate the electric field (in volts per metre) experienced by the anion when the...
-
Which of the following molecules may be polar: CIF 3 , O 3 , H 2 O 2 ?
-
If X is a random variable having a normal distribution with the mean and the standard deviation s, use the third part of Theorem 4.10 on page 128 and Theorem 6.6 to show that the moment generating...
-
What will happen to a country that fixes the price of the foreign exchange below equilibrium? What factors will shift the supply and demand for currency?
-
paid $ 8 2 on January 2 2 0 2 4 , for 2 shares of Came Cosmetics common stock The investment represents a 2 0 % interest in the net assets of Came and gave the ability to exercise significant...
-
Discuss the economics of free 2 year education at community college for all Americans. Can you relate this to grade inflation?
-
A good investor is also a good arbitrageur who keeps constant awareness on chances to make easy money. Suppose you observe the following exchange rates: S($/) = $1.3/. The one-year forward rate is F...
-
?prepare a journal entry for 1 ) ?direct materials 2 ) ?direct labor and 3 ) ?manufacturing overhead costs 13 Job No. Direct Materials Direct Labor 50 $11,500 $5,750 51 44,850 28,750 52 34,500 23,000
-
List five factors that control when and if a cell divides.
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
The centre of the EPR spectrum of atomic deuterium lies at 330.02 mT in a spectrometer operating at 9.2482 GHz. What is the g-value of the electron in the atom?
-
A radical containing three equivalent protons shows a four-line spectrum with an intensity distribution 1:3:3:1. The lines occur at 331.4 mT, 333.6 mT, 335.8 mT, and 338.0 mT. What is the hyperfine...
-
A radical containing three in equivalent protons with hyperfine constants 2.11 mT, 2.87 m'T and 2.89 mT gives a spectrum centred on 332.8 mT. At what fields do the hyperfine lines occur and what are...
-
What is the yield to maturity of a 23-year bond that pays a coupon rate of 8.25% per year, has a $1,000 par value , and is currently priced at $1,298.05?
-
Suppose the exchange rate between U.S. dollars and Swiss francs is SF 1.112 = $1.00, and the exchange rate between the U.S. dollar and the euro is $1.00 = 0.9842 euros. What is the cross-rate of...
-
Use the following information for the Lowell, Inc. for this and the next two questions. Sales $200,000 Debt 95,000 Dividends 5,000 Equity 40,000 Net income 16,000 1.What is the company's...

Study smarter with the SolutionInn App


