The table below lists the vapour pressures of mixtures of iodoethane (I) and ethyl acetate (A) at
Question:
The table below lists the vapour pressures of mixtures of iodoethane (I) and ethyl acetate (A) at 50°C. Find the activity coefficients of both components on
(a) The Raoult’s law basis,
(b) The Henry’s law basis with iodoethane as solute.
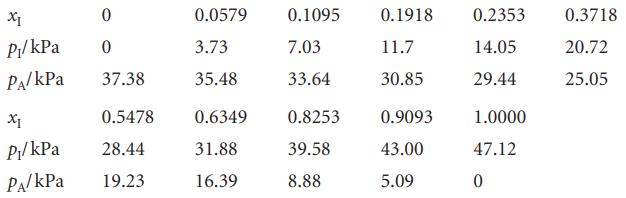
Transcribed Image Text:
XI 0 P₁/kPa 0 PA/kPa 37.38 X1 0.5478 P₁/kPa 28.44 PA/KPa 19.23 0.0579 3.73 35.48 0.6349 31.88 16.39 0.1095 7.03 33.64 0.8253 39.58 8.88 0.1918 11.7 30.85 0.9093 43.00 5.09 0.2353 14.05 29.44 1.0000 47.12 0 0.3718 20.72 25.05
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The table below lists gross domestic product (GDP), consumption (C), gross private domestic investment (I), government spending (G), and net exports (X M). Compute each as a percent of GDP for the...
-
The table below lists the cost per serving, in dollars, for items on four menus that are served at an elder-care facility. On a particular day, a dietician orders 32 meals from menu 1, 19 from menu...
-
The table below lists the cost per serving, in dollars, for items on three lunch menus served at a senior citizens' center. On a particular day, 26 Menu 1 meals, 18 Menu 2 meals, and 23 Menu 3 meals...
-
Willingham Company Ltd. has the following comparative statements of financial position data. WILLINGHAM COMPANY LTD. Statements of Financial Position December 31 Additional information for 2017: 1....
-
Compute the present value of the preceding cash flows.
-
Oscillations of a Piston A vertical cylinder of radius r contains a quantity of ideal gas and is fitted with a piston with mass m that is free to move (Fig. 19.34). The piston and the walls of the...
-
Refer to Exercise 9. Can you reject H0 at the = 0.01 level? State a conclusion. Exercise 9 Two suppliers of machine parts delivered large shipments. A simple random sample of 150 parts was chosen...
-
Newland Company reported retained earnings at December 31, 2016, of $310,000. Newland had 200,000 shares of common stock outstanding at the beginning of 2017. The following transactions occurred...
-
An investment Advisor has a portfolio worth $60 Million with a beta of 0.75. The manager is concerned about the performance of the market over the next 2 months and plans to use 3-month futures...
-
In the United States, the accounting profession is regulated at the state level, while in India the accounting profession is regulated by a federal agency. Identify and briefly discuss the...
-
Consider a container of volume 5.0 dm 3 that is divided into two compartments of equal size. In the left compartment there is nitrogen at 1.0 atm and 25C; in the right compartment there is hydrogen...
-
The osmotic pressure of an aqueous solution at 300 K is 120 kPa. Calculate the freezing point of the solution.
-
Express cos(/2 + 2x) in terms of sin x and cos x.
-
The map above shows a river and its floodplain. The heavy dashed lines mark the edges of the 25-year floodplain and the light dashed lines mark the edges of the 5-year floodplain. The letter "X" lies...
-
Beginning inventory, purchases, and sales for Item CSW15 are as follows: August 1 Inventory 102 15 9 Sale 90 16 Purchase 126 27 25 Sale 78 Assuming a perpetual inventory system and using the...
-
. For each case, determine the Resultant Moment about point A produced by the forces. A 2000 lb 60 B 800 lb A 60 5" - 30 mm A B C 2 m 36" (a) 50 3 kN 0 (b) 13 5 B 12 5 kN 4' 10' (c)
-
Discuss three strategies that a leader can use for building trust and confidence with colleagues during times of organisational change.
-
RealityView makes augmented reality goggles at low cost. The company has a plant capacity of 200,000 units. Variable costs are $4,000,000 at 100% capacity. Fixed costs are $2,000,000 per year, but...
-
Why do you think 'Nicky's Rules' help to make the Tea and Sympathy operation more efficient?
-
A simple random sample of 220 university students were asked what pasta they usually order and with which sauce. The preferences of these respondents are summarised below: Sauce Bolognese Pasta...
-
Suppose that a molecular orbital of a heteronuclear diatomic molecule is built from the orbital basis A, B, and C, where B and C are both on one atom (they can be envisaged as F2s and F2p in HF, for...
-
Sketch the molecular orbital energy level diagram for IF and deduce its ground-state electron configuration. Is IF likely to have a shorter bond length than IF or IF + ?
-
Why do both ionization energy and electron affinity play a role in estimating the energy of an atomic orbital to use in a molecular structure calculation?
-
Firm A invents a new product and obtains a patent for it. It calls the product "Asky". Because of the patent, only Firm A can produce and sell Asky. Firm A plans to sell 1,000 units of Asky at the...
-
You are the Senior Manager of IAuditYou LLP, you were recently assigned to take over a very important client for the company, The engagement partner, Max Roff, has been the audit partner for the past...
-
The issues we face towards building a sustainable and just world run according to the principles by Ostrom are due to several factors but mainly the desire for humanity to develop economically at all...

Study smarter with the SolutionInn App


