The unimolecular decomposition of urea in aqueous solution is measured at two different temperatures and the following
Question:
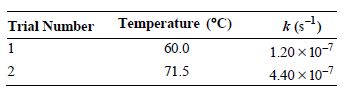
a. Determine the Arrhenius parameters for this reaction.
b. Using these parameters, determine ΔH€¡ and ΔS€¡ as described by the Eyring equation.
Transcribed Image Text:
Temperature (°C) k (s-) Trial Number 60.0 1.20 x 10-7 71.5 4.40 x 10-7 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a The activation energy is determined by taking the ratio of the rate constants ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Hydrogen abstraction from hydrocarbons by atomic chlorine is a mechanism for Cl · loss in the stratosphere. Consider the reaction of Cl· with ethane: C 2 H 6 (g) + Cl· (g) C 2 H...
-
Suppose the vapor pressure of a substance is measured at two different temperatures. (a) By using the Clausius-Clapeyron equation (Equation 11.1) derive the following relationship between the vapor...
-
The gas-phase decomposition of ethyl bromide is a first-order reaction, occurring with a rate constant that demonstrates the following dependence on temperature: a. Determine the Arrhenius parameters...
-
why is teamwork so important especially in healthcare? explain
-
Find (a) the domain and (b) the zeroes of the function. 1. f (x) = (x -5) / (2x2 - x) 2. f (x) = 10 - (3 - x)
-
A small coil, of area \(2 \mathrm{~cm}^{2}\), carries a current of \(1.5 \mathrm{~A}\). The coil is suspended inside a solenoid at an angle of \(45^{\circ}\) with respect to the magnetic field. The...
-
A fixed mass of gas emits \(250 \mathrm{~J}\) of heat energy and contracts at a constant pressure of \(1 \times 10^{5} \mathrm{~Pa}\) from \(2.5 \times 10^{-3} \mathrm{~m}^{3}\) to \(1.0 \times...
-
Morettis Frozen Pizza Inc. has determined from its production budget the following estimated production volumes for 12'' and 16'' frozen pizzas for September 2014: There are three direct materials...
-
A particle undergoes damped harmonic motion. The spring constant is 100 N/m; the damping constant is 8.0 x 10-3 kg?m/s, and the mass is 0.050 kg. If the particle starts at its maximum displacement, x...
-
A ride hailing company has their DB structured in 3 major tables as described in the SCHEMA section below. Write a query to fetch the top 100 users who traveled the most distance using the service....
-
Catalase is an enzyme that promotes the conversion of hydrogen peroxide (H 2 O 2 ) into water and oxygen. The diffusion constant and radius for catalase are 6.0 10 7 cm 2 s 1 and 51.2 ,...
-
Consider the unimolecular isomerization of methylcyanide, a reaction that will be discussed in detail in Chapter 36: CH 3 NC(g) CH 3 CN(g) The Arrhenius parameters for this reaction are A = 2.5 10...
-
The United Transportation Workers went on strike after their contract expired with Safeway Trails Bus Company. The company submitted to the union a new proposal for ratification. John Lantz, the...
-
what ways does the concept of biocultural diversity broaden our understanding of the relationship between human societies and biodiversity, and how can this concept inform more holistic conservation...
-
The force of interest is defined as follows, 2 8(t) = = t> 0 3(1+t)' By using the above force of interest, answer the following questions:- a. Formulate a(t). b. Determine i3. c. Evaluate the present...
-
President Wilson quite clearly distinguishes between politics and administration in his piece. He claims that they are two different things. There are those who are politicians and those who are...
-
What types of assets should be purchased to support new projects and generate future cash flows? The stock market is approaching its year-to-date high value, so should Phoenix Golf Club Company...
-
In referencing The Immortal Life of Henrietta Lacks. How did the Lacks family, the media, and the general population view the medical community during the 1950s? What is the Nuremberg Code? How was...
-
Graph 2 f(x) = 3 [3 if x + 2 if x = 2
-
Identify the Critical Infrastructure Physical Protection System Plan.
-
At 298 K and 1 bar pressure, the density of water is 0.9970 g cm 3 , and C P,m = 75.3 J K -1 mol -1 . The change in volume with temperature is given by V = V i T, where , the coefficient of thermal...
-
5-Methylcyclopentadiene undergoes homolytic bond cleavage of a C!H bond to form a radical that exhibits five resonance structures. Determine which hydrogen is abstracted and draw all five resonance...
-
Calculate S surroundings and S total for the processes described in parts (a) and (b) of Problem P5.16. Which of the processes is a spontaneous process? The state of the surroundings for each part is...
-
Use the table for the question(s) below. Suppose the term structure of interest rates is shown below: Term Rate 1 year 2 years 3 years 5 years 10 years 20 years (EAR%) 5.00% 4.80% 4.60% 4.50% 4.25%...
-
Alpha and Beta Companies can borrow for a five-year term at the following rates: Alpha Beta Moody's credit rating Aa Baa Fixed-rate borrowing cost 10.5% 12.0% Floating-rate borrowing cost LIBOR...
-
4) Transactions for the month of July for Shelly's Bookstore were as follows: Purchases Salca July 1 (balance) 600 8 $7.30 1,200 @ $7.40 July 3 350 @$9.50 12 900 @9.75 22 700 $7.60 25 800 10.00 250...

Study smarter with the SolutionInn App


