The viscosity of benzene varies with temperature as shown in the following table. Use the data to
Question:
The viscosity of benzene varies with temperature as shown in the following table. Use the data to infer the activation energy for viscosity.
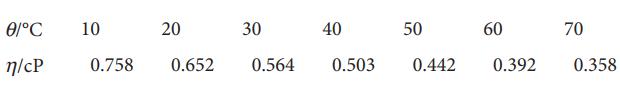
Transcribed Image Text:
Ꮎ/l°C n/cP 10 0.758 20 30 40 50 0.652 0.564 0.503 0.442 60 0.392 70 0.358
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
The activation energy for viscosity can be estimated using the Arrhenius equation ln0 ...View the full answer

Answered By

Dansteve Matoke
As a consequence of more than three years of experience in ACADEMIC WRITING, I have vast, diverse knowledge and impeccable grammar. I guarantee the quality of my work across multiple fields. I intend to precisely meet the clients' expectations. Let's work together to achieve exceptional grades.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
The molar heat capacity of lead varies with temperature as follows: TIK 10 15 20 25 30 50 Cp,m/O K-1 rnol-1) 2.8 7.0 10.8 14.1 16.5 21.4 TIK 70 100 150 200 250 298 Cp,m/OK-1 mol-1) 23.3 24.5 25.3...
-
The data shown in the following table were obtained from the 1990 Census. Included is information on 26 randomly selected Metropolitan Statistical Areas (MSAs). Of interest are factors that...
-
The data shown in the following table are highway gasoline mileage performance and engine displacement for a sample of 20 cars. (a) Fit a simple linear model relating highway miles per gallon (y) to...
-
A horizontal cantilever beam of length l and of uniform cross-section carries a uniformly distributed load of w per unit length for the full span. The cantilever is supported by a rigid prop at a...
-
A woman with epilepsy worked as a regional medical director for an insurance company. Following a seizure, she was banned from driving for six months by her doctor. She requested that she be allowed...
-
Why would a company maintain separate accounts receivable ledgers for each customer, as opposed to maintaining a single accounts receivable ledger for all customers?
-
In Example 13.5, we constructed a 95% confidence interval for the slope 1 in the model to predict the number of calories from the number of grams of fat. The 95% confidence interval is 4.8182 y|20 be...
-
Valuing a right Knight inventory Systems, Inc., has announced a rights offer. The company has announced that it will take four rights to buy a new share in the offering at a subscription price of...
-
Task 2: Return, Risk and the Security Market Line wwwwww wwwww Suppose, you have just begun working for an investment firm called Covili and Wyatt. Paul Covili, one of the firm's founders, has been...
-
An empirical expression that reproduces the viscosity of water in the range 20100 C is where 20 is the viscosity at 20C. Explore (by using mathematical software) the possibility of fitting an...
-
Calculate the thermal conductivity of argon (C V,m =12.5 JK 1 mol 1 , =0.36nm 2 ) at 298K.
-
Explain how Ficks first law arises from the concentration gradient of gas molecules.
-
We would like to produce a 2-in.-thick steel wear plate for a rock-crushing unit. To avoid frequent replacement of the wear plate, the hardness should exceed HRC 38 within 0.25 in. of the steel...
-
Introduction: Top Secret, Inc. (TSI) faces significant business-related challenges stemming from the use of a single-purpose operating system (TSI OS) for its back-office functions. This decision,...
-
Suppose you, as a college student, could choose between completing either a math assignment or a microeconomics assignment. Assume that you could complete the math assignment in 30 minutes, but...
-
1. How can you use current and emerging sector developments and/or research to improve practice? Describe an example that demonstrates this, 2. Suggest two occasions when specialist advice or the...
-
Calculate the amount of cash collections you will collect in November if you estimate you will collect 60% of your sales in the month of the sale, 25% the following month, 10% two months later and...
-
Describe the five characteristics of life, and list severalnonliving things that possess at least two of thesecharacteristics. Explain how an atom relates to the biosphere by describing thelevels of...
-
Spring Street Engine Shop uses a job order cost system to determine the cost of performing engine repair work. Estimated costs and expenses for the coming period are as follows: Engine parts...
-
2.) Find the Laplace transform of f(t) 7e-St cos 2t +9 sinh2 2t. Use Laplace Table. %3D
-
A surface displaying a contour of the total charge density in LiH is shown here. The molecular orientation is the same as in Problem P23.13. What is the relationship between this surface and the MOs...
-
Show that the Slater determinant formalism automatically incorporates the Pauli exclusion principle by evaluating the He ground-state wave function of Equation (21.9), giving both electrons the same...
-
Identify the molecular orbitals for F 2 in the images shown here in terms of the two designations discussed in Section 23.7. The molecular axis is the z axis, and the y axis is tilted slightly out of...
-
cos (2x2)-1 Let f(z) = Evaluate the 6th derivative of fat z = 0. 2 f(0) (0)= Hint: Build a Maclaurin series for f(x) from the series for cos(x).
-
Do you think that it should be legal to falsely implicate a suspect as a participant in a crime by suggesting that there is physical evidence from their person, reflecting that they were present at...
-
In this practice activity, you will identify key roles and responsibilities of criminal justice professionals regarding victims. You will also describe potential consequences to victims when those...

Study smarter with the SolutionInn App


