Use standard Gibbs energies of formation to calculate the standard reaction Gibbs energies at 298K of the
Question:
Use standard Gibbs energies of formation to calculate the standard reaction Gibbs energies at 298K of the reactions
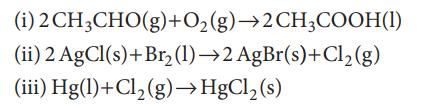
Transcribed Image Text:
(i) 2 CH3CHO(g) + O₂(g) →2CH3COOH(1) (ii) 2 AgCl(s) + Br₂ (1)→2 AgBr(s)+Cl₂(g) (iii) Hg(1)+Cl₂(g) →HgCl₂ (s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Answer and thorough explanation For reaction i we ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Use standard Gibbs energies of formation to calculate the standard reaction Gibbs energies at 298 K of the reactions in Exercise 3.8b.
-
Sandesh and Rani are the only directors and shareholders of SaR Pty Ltd. SaR owns several tenanted properties. Most of the properties are commercial buildings, but it also owns some residential...
-
Use the tabulated free energies of formation to calculate the equilibrium constant for the reaction at 298 K: H 2 (g) + Cl 2 (g) 2HCl (g)
-
Subsequent Events and Subsequently Discovered Facts. Michael Ewing is auditing the financial statements of Dallas Company for the year ended December 31, 2014. In concluding the process of gathering...
-
On January 1, 2015, the Tara Clothing Corporation issued $ 900,000 par value, 5%, six- year bonds. Interest is payable semiannually each January 1 and July 1 with the first interest payment due at...
-
A voluntary health and welfare organization summarizes its expenses by function, as follows: Education........ $20,400 Fund-raising...... 11,400 Management and general.. 5,500 Public health.........
-
How can a paralegal assist a lawyer with the voir dire of the jury?
-
Enter the following column headings across the top of a sheet of paper: Enter the transaction letter in the first column and show the effect (if any) of each of the following transactions on each...
-
Give a article in any publication that discusses unusual, or interesting forecasting method or use of forecasting a business operation Then summarize the article you found Provide a link to or...
-
M (a) Find the time path of capital K(t) given the following rates of net investment flow functions I(t) = 30t/2 K(0) = 100 I(t) = 7t/2 (2 Marks) (2 Marks) I(t) = 50t/4 (2 Marks) For each of (i) to...
-
Discuss the relationships between the various formulations of the Second Law of thermodynamics.
-
Use mathematical software, a spreadsheet, or the Living graphs on the web site for this book to: (a) Evaluate the change in entropy of 1.00mol CO 2 (g) on expansion from 0.001m 3 to 0.010m 3 at 298K,...
-
A consumer price analyst claims that prices for liquid crystal display (LCD) computer monitors are normally distributed, with a mean of $190 and a standard deviation of $48. What is the probability...
-
A particle initially has a speed of 0.46c. (Enter your answers in terms of c. Round your answers to at least three decimal places.) (a) At what speed does its momentum increase by 1%? .465 x c (b) At...
-
Explain in detail, in the UK the need to reassure children, young people and adults of the confidentiality of shared information and the limits of this give reference and examples ?
-
The "one-time good deal" Cash-For-Clunkers program offered by the federal government proved a temporary boon for car dealers. In addition to this program, dealers were eager to add their own...
-
Solve: 3a+11 = 2. (Use a comma to separate multiple solutions. Enter if there is no real solution.) Provide your answer below:
-
solve x-x-12=0
-
Suppose u1,.......,uk form an orthonormal set of vectors in Rn with k < n. Let Q = (u, u2 ... uk) denote the n k matrix whose columns are the orthonormal vectors. (a) Prove that QT Q = Ik. (b) Is...
-
The roof of a refrigerated truck compartment is of composite construction, consisting of a layer of foamed urethane insulation (t2 = 50 mm, ki = 0.026 W/m K sandwiched between aluminum alloy panels...
-
Discuss the steps involved in the calculation of the energy of a system by using the variation principle.
-
Hydrogen is the most abundant element in all stars. However, neither absorption nor emission lines due to neutral hydrogen are found in the spectra of stars with effective temperatures higher than 25...
-
Give the possible term symbols for (a) Li [He]2s 1 , (b) Na [Ne]3p 1 .
-
This course was very challenging and engaging. I really enjoyed everything that I learned during this course that will help me run a successful business. What have you learned in this course and...
-
Complete the income statement for the following transactions for June 2010 disclosed Fill in the yellow cells as appropriate. Ticket revenue $180,000 Equipment rent expense $50,000 Film rent expense...
-
Westerville Company reported the following results from last year's operations: \ table [ [ \ table [ [ Sales ] , [ Variable expenses ] , [ Contribution margin ] , [ Fixed expenses ] ] , $ 1 , \...

Study smarter with the SolutionInn App


