Use the result of Problem P3.10 to derive a formula for (CV /V ) T for a
Question:
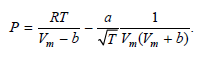
Transcribed Image Text:
RT 1 a Vm - b VT VVm + b)' T VVm P:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
We use the relationship ...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the result of Problem P3.10 to show that (C V /V) T for the van der Waals gas is zero.
-
Use the result of Problem 21 to calculate the total translational kinetic energy of the conduction electrons in 1.00 cm3 of copper at T = 0 K.
-
In this problem, you are to use the result of Problem 129 to derive Equation 14-45, which relates the width of the resonance curve to the Q value when the resonance is sharp. At resonance, the...
-
Suppose you are going to put up your own company, list down at least ten of your possible contributions to the socio-economic development in your community. Name of Business: Name of Owner/s: 2. 4....
-
Make a presentation on "Ken Chenault", Transforming the Leadership Landscape
-
A car salesman meets 12 customers each week. His probability of making a sale in the first meeting with a customer is .3. When he gets the second meeting with the same customer, the probability of...
-
The following transactions occurred between Kinzer Furniture and M & L Furniture Warehouse during May of the current year: Requirements 1. Journalize these transactions on the books of Kinzer...
-
A vacuum system, as used in sputtering electrically conducting thin films on microcircuits, is comprised of a base plate maintained by an electrical heater at 300 K and a shroud within the enclosure...
-
You have been provided the following financial statements for Big XTC Company. Balance Sheet (5000's) Forecast ASSETS 2018 2019 2020 Current Assets Cash 9,000 7,282 14,000 Short-term Investments...
-
Michigan Motors Corporation (MMC) just introduced a new luxury touring sedan. As part of its promotional campaign, the marketing department decided to send personalized invitations to test-drive the...
-
Karen runs a print shop that makes posters for large companies. It is a very competitive business. The market price is currently $1 per poster. She has fixed costs of $250. Her variable costs are...
-
Relate opportunity costs to why profits encourage entry into purely competitive industries and how losses encourage exit from purely competitive industries.
-
What does it mean to use relative performance evaluations when assessing the performance of a subunit?
-
Have you experienced Simpson's paradox in your professional and/or personal life? If so, please describe the context in which it occurred.
-
Assess the models in Problems 9.12 and 9.14 Problem 9.12 is: and Problem 9.14 is: . 9.12 Generalize the model considered in Example 4.11 to a marginal model for the longi- tudinal DOS data and...
-
A random sample of 16 subjects was taken from a target population to study the prevalence of a disease \(p\). It turned out that six of them were diseased. (a) Estimate the disease prevalence \(p\)....
-
Consider the statistic in (2.14). (a) Show that this statistic is asymptotically normal with the asymptotic variance given by...
-
The data set "intake" contains baseline information of the Catheter Study. Use the two binary outcomes on whether urinary tract infections (UTIs) and catheter blockages occurred during the last two...
-
The fixed budget performance report for the year ended 30 June 2018 for Motueka Mint is as follows: Budget Actual Variance Units of production: 84 000 94 000 10 000 F Factory overhead: Variable...
-
The following selected accounts and normal balances existed at year-end. Notice that expenses exceed revenue in this period. Make the four journal entries required to close the books: Accounts...
-
A sample of 1.00 mol perfect gas molecules with Cp,m = 7/2 R is put through the following cycle: (a) Constant-volume heating to twice its initial volume, (b) Reversible, adiabatic expansion back to...
-
The molar heat capacity of ethane is represented in the temperature range 298 K to 400 K by the empirical expression Cp,m/ (J K-1 mol-1) = 14.73 + 0.1272(T/K). The corresponding expressions for C(s)...
-
The standard enthalpy of formation of the metallocene bis (benzene) chromium was measured in a calorimeter. It was found for the reaction Cr (C6H6)2(s) Cr(s) + 2 C6H6 (g) that Uo (583 K) = +8.0 kJ...
-
For 2012, Tri-Comic Company initiated a sales promotion campaign that included the expenditure of an additional $50,000 for advertising. At the end of the year, Lumi Neer, the president, is presented...
-
Initially at rest, an egg is released from the top of a tall building. After a brief delay, a second egg is hurled down from the same height at a rate of 6.15 m/s. If both eggs splat on the ground at...
-
(a) Can a single isolated stationary object of mass 4 spontaneously explode into two objects each of rest mass greater than 2? Explain your reasoning. (b) Can a single isolated stationary object of...

Study smarter with the SolutionInn App


