Using the formula for the energy levels for the Morse potential, show that the energy spacing between
Question:
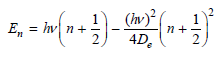
show that the energy spacing between adjacent levels is given by
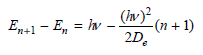
Transcribed Image Text:
(hv)² n + E, = hv n+ ADE En+1 - E, = iw 2D. (in² (n + 1) (Iv)?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
Eus E tv x 2 x x x 1 hvn 3 hv ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using the Bohr formula for the energy levels, calculate the energy required to raise the electron in a hydrogen atom from n = 1 to n = . Express the result for 1 mol H atoms. Because the n = level...
-
Calculate the ionization energy of the He+ ion in kJ/mol (this would be the second ionization energy of He). See Problem 8.107. The Bohr formula for the energy levels of an ion consisting of a...
-
Using the formula for the moment of inertia of a uniform sphere, find the moment of inertia of a thin spherical layer of mass m and radius R relative to the axis passing through its centre.
-
What are four advantages of integrating AI technologies into decision support systems?
-
Consider the function f (x) = 3 sin (0.6x 2). (a) Approximate the zero of the function in the interval [0, 6]. (b) A quadratic approximation agreeing with f at x = 5 is g (x) = -0.45x2 + 5.52x -...
-
Specifications on a cigar lighter detent are 0.3220 and 0.3200 in. Samples of size 5 are taken every 45 minutes with the results shown in Table 6E.18 (measured as deviations from 0.3210 in 0.0001...
-
Two balls are drawn from a bag containing 20 white and 5 black balls. In the first draw a ball is drawn at random and then replaced in the bag. In the second, a ball is drawn again at random. Find...
-
Zimmerman Clothiers manufactures womens business suits. The company uses a standard cost accounting system. In March 2014, 15,700 suits were made. The following standard and actual cost data applied...
-
What is the prevalence of Forensic Psychology? Explain the purpose of Forensic Psychology in what unique assessments are provided?
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
A measurement of the vibrational energy levels of 12 C 16 O gives the relationship where n is the vibrational quantum number. The fundamental vibrational frequency is ν 0 = 2170.21 cm...
-
Use your results from P19.36 to solve the following problem. For 1 H 35 Cl, D e = 7.41 10 -19 J and = 8.97 10 13 s -1 . As n increases, the energy difference between adjacent vibrational levels...
-
Show that the given families of curves are orthogonal trajectories of each other. Sketch both families of curves on the same axes. 59. x + y? = r?, ax + by = 0 60. x + y - ax, x + y? = by 61. y = ex,...
-
Assume that the monthly payments faced by an insurance company are normally distributed with a mean of \($100,000\) and a standard deviation of \($12,000.\) Determine the probability that in a given...
-
Suppose you manage a pension fund and an investment bank contacts you to review the prospectus of a security issue that you know is currently undersubscribed. Assuming your research indicates that...
-
Bluestar Airlines has physical assets valued at \($12,500,000,\) and according to industry experts, the net present value of its future earnings amounts to \($40.00\) per share. Assuming there are...
-
Explain the working principle of the open cycle gas turbine.
-
Xavi decides to short sell 10,000 shares of Guiness Co. stock, currently trading at \($7.00\) per share. Determine Xavis profits if Guiness stock trades at \($5.00\) per share at the moment in which...
-
How are the derivatives of sin -1 x and cos -1 x related?
-
Periwinkle Company is a multinational organization. Its Parts Division is located in Lavender Land, while its Assembly Division is located in North Orchid. During the current year Periwinkle Companys...
-
Air in industrial plants is subject to contamination by many different chemicals, and companies must monitor ambient levels of hazardous species to be sure they are below limits specified by the...
-
An adult takes about 12 breaths per minute, inhaling roughly 500 mL of air with each breath. The molar compositions of the inspired and expired gases are as follows: The inspired gas is at 24C and 1...
-
A spray-drying operation similar to that described in Problem 5.23 is used by the pharmaceutical industry. Acetaminophen is an active pharmaceutical ingredient (API) that can be produced in powdered...
-
Consider the stock of Aya Company, which will pay an annual dividend of $3 one year from today. The dividend will grow at a constant rate of 3%, forever.The market requires a 12% return on the...
-
Analyze the video presented on the link below. Also, please discuss or explain the video and relate it to personal finance. https://www.youtube.com/watch?v=nFY0HBkUm8o
-
After the company issues an initial public offering and they desire to offer additional shares, one option is the seasoned equity offering. What are the differences between a convertible bond and a...

Study smarter with the SolutionInn App


