What can you say about the composition of the solid below the eutectic temperature in Figure 9.26
Question:
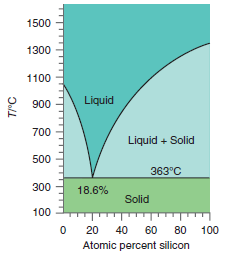
Transcribed Image Text:
1500 1300 1100 Liquid 900 700 Liquid + Solid 500 363°C 300 18.6% Solid 100 20 40 60 80 100 Atomic percent silicon T/°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 38% (13 reviews)
Gold and silicon are not miscible in all propo...View the full answer

Answered By

Manish Sahu
I worked some of the coaching institute of CSIR NET,GATE in Bhubaneswar and my students are really doing good and response from there side is positive.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Three particles, a, b, and c, enter a magnetic field as shown in Fig. 20-46, what can you say about the charge on each particle? a, b. c b.
-
Two different gases are at the same temperature. What can you say about the rms speeds of the gas molecules? What can you say about the average kinetic energies of the molecules?
-
Two constant-volume stirred tanks, connected in series (see Figure a), are fed by a single stream with constant volumetric flow rate, q. The feed composition, c1 (mass/volume of a catalyst species),...
-
Go out onto the Web and compare three shopping bots for a product you are interested in (e.g., www.mysimon.com, www.bottomdollar.com, www.shopzilla.com, www.shopping.com, or www.pricegrabber.com)....
-
The following information relates to next year's projected operating results of the Aluminum Division of DG Corporation: Contribution margin ............... $1,500,000 Fixed...
-
Why do we need to distinguish capital gains separately from other income?
-
Raoult's law is found to be quite satisfactory for a (a) Dilute solution (b) Ideal solution (c) Non-ideal solution (d) Perfect gas.
-
A mining company is deciding whether to open a strip mine, which costs $2 million. Net cash inflows of $13 million would occur at the end of Year 1. The land must be returned to its natural state at...
-
How does this portrayal of the legislative process present anoversimplified journey by which a bill becomes a law? What elsewould need to be addressed to make this portrayal moreaccurate?
-
1. Measuring and monitoring retention and turnover data traditionally involved assessing total turnover. Many organizations fail to analyze retention beyond this number. Describe a thorough approach...
-
You boil an ethanolbenzene mixture with x ethanol = 0.35. What is the composition of the vapor phase that first appears? What is the composition of the last liquid left in the vessel?
-
The statement The boiling point of a typical liquid mixture can be reduced by approximately 100C if the pressure is reduced from 760. to 20. Torr is found in Section 9.4. What figure(s) in Chapter 9...
-
Tech is playing State in the last conference game of the season. Tech is trailing State 21 to 14 with 7 seconds left in the game, when they score a touchdown. Still trailing 21 to 20, Tech can either...
-
The Cutting Department of Lasso Company has the following production and cost data for August. Production Costs 1. Started and completed 9,600 units. Beginning work in process $0 2. Started 2,300...
-
Cali Windows is a small company that installs windows. Its cost structure is as follows: (Click the icon to view the cost structure.) Calculate (a) the breakeven point in units and revenues and (b)...
-
Determine if the following values are zeroes polynomial. 4 3 f(x) = x + x-18x - 16x + 32 x+1, +2, +4 -
-
Summarize each data source and include them for substance abuse and alcohol in the military. Analyze each data source for substance abuse and alcohol in the military for trustworthiness and accuracy....
-
5 5 Color the World Co., with a December 31 year end date, started to sell high definition televisions for $2,000 each on Jan 1, 2018. Each television includes one year warranty covering parts and...
-
If /4 0 tan 6 x sec x dx = I, express the value of /4 0 tan 8 x sec x dx in terms of I.
-
Fred Farmer needs to prepare a balance sheet for his bank. He spent the day getting the following information. Fred needs your help to build a balance sheet and evaluate it. The information was...
-
In the cryoscopic determination of molar mass, the molar mass in kg mol 1 is given by where W is the mass of the solvent, w is the mass of the unknown solute, ÎT f is the amount by which the...
-
The following data give the vapor pressure of water at various temperatures. Transform the data, using ln (P) for the dependent variable and 1/T for the independent variable. Carry out the linear...
-
The following is a set of student data on the vapor pressure of liquid ammonia, obtained in a physical chemistry laboratory course. (a) Find the indicated enthalpy change of vaporization. Temperature...
-
Using the car mileage data from Lab 1, define the following sets: A = {x|x> 125,000), B = {xx < 175,000) and C = {xlx <50,000). Demonstrate the following set equivalencies (DeMorgan's Law) using R...
-
6. Write out the truth tables for: a) a 3 input and gate b) an exclusive or gate c) a two input inclusive or gate 3 7. Describe the microprocessor instruction you would use to: 8. a) set bit 4 of a...
-
Alex Corporation reports the following components of stockholders' equity at December 31 of the prior year. Common stock-$25 par value, 50,000 shares authorized, 33,000 shares issued and outstanding...

Study smarter with the SolutionInn App


