Why are there no points in the phase diagram for sulfur in Figure 8.11 that show rhombic
Question:
Figure 8.11
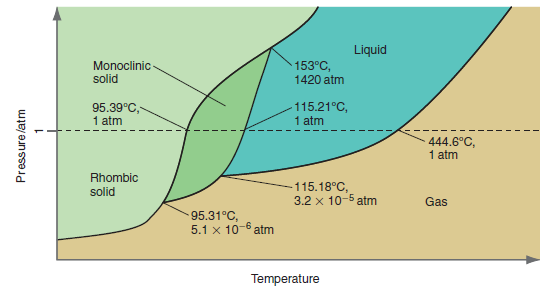
Transcribed Image Text:
Liquid Monoclinic- solid 153°C, 1420 atm 95.39°C, 1 atm 115.21°C, 1 atm 444.6°C, 1 atm Rhombic -115.18°C, 3.2 x 10-5 atm solid Gas 95.31°C, 5.1 x 10-6 atm Temperature Pressure/atm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (18 reviews)
According to the ...View the full answer

Answered By

Ashok Kumar Malhotra
Chartered Accountant - Accounting and Management Accounting for 15 years.
QuickBooks Online - Certified ProAdvisor (Advance - QuickBooks Online for 3 years.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The phase diagram of NH 3 can be characterized by the following information. The normal melting and boiling temperatures are 195.2 and 239.82 K, respectively; the triple point pressure and...
-
A PT phase diagram for potassium is shown below. a. Which phase has the higher density, the fcc or the bcc phase? Explain your answer. b. Indicate the range of P and T in the phase diagram for which...
-
Why are there no sp4 or sp5 hybrid orbitals?
-
The contingency table shown relates happiness and gender for the 2012 GSS. a. Identify the response variable and the explanatory variable. b. Construct a table or graph showing the conditional...
-
1. Draw the appropriate network diagram. 2. Find the Critical Path and the project completion time. 3. Find the Probability that the project will take more than 50 days to complete. Please use excel...
-
What is a hypothesis test?
-
An integration method in which the computation of \(x_{i+1}\) is based on the equilibrium equation at \(t_{i}\) is known as a. explicit method b. implicit method c. regular method
-
Sharkeys Fun Center contains a number of electronic games as well as a miniature golf course and various rides located outside the building. Paul Sharkey, the owner, would like to construct a water...
-
Balance, September 1 Checks BANK STATEMENT Deposits Other Balance $ 1,940 September 7 NSF check $ 90 1,850 September 11 $ 2,910 4,760 September 12 #101 $ 770 3,990 September 17 #102 1,640 2,350...
-
Robert Gates rounds the corner of the street and smiles when he sees his wife pruning rose bushes in their front yard. He slowly pulls his car into the driveway, turns off the engine, and falls into...
-
Calculate the degree of dissociation of N 2 O 4 in the reaction N 2 O 4 (g) 2NO 2 (g) at 300. K and a total pressure of 1.50 bar. Do you expect the degree of dissociation to increase or decrease as...
-
Predict which of the following substrates will undergo an E1 reaction more quickly. Explain your choice. Br Br or
-
Find the ratio of Earth's gravitational force on a satellite when it is on the ground to the gravitational force exerted when the satellite is orbiting at an altitude of 320 km.
-
A speculator owns a portfolio of options on Apple stock. The speculator is long 15 call option contracts with a strike price of $170 per share, the speculator is short 30 call option contracts with a...
-
How can I get the refund for the last payment as I am not going to need the access as I graduated already?
-
Stock Valuation using a Dividend Discount Model Roadrunner Enterprises is expected to grow its dividends and earnings at various rates. The company just paid a cash dividend of $3.00 per share. The...
-
Building a blank can be the most blank of writing your business plan. It's also one of the most blank . Businesses that have a blank in place more prepared to pitch to investors, receive funding, and...
-
Annotate two points of interest from your readings this week. Please respond to at least 3 other students. Responses can include direct questions. Note: Please bear in mind throughout the coursethe...
-
Evaluate S 10 , the sum of the first ten terms, for the arithmetic sequence. 8, 11, 14, . . .
-
Determine the resultant moment produced by the forces about point O. 0.25 m 0.125 m, 0 0.3 m- 60 F = 500 N F = 600 N
-
A gas obeying the equation of state p(V-nb) = nRT is subjected to a Joule- Thomson expansion. Will the temperature increase, decrease, or remain the same?
-
Rearrange the van der Waals equation of state to give an expression for T as a function of p and V (with n constant). Calculate (T/p)v and confirm that (T/p)v= l/(p/D")v. Go on to confirm Euler's...
-
On a cold, dry morning after a frost, the temperature was -5C and the partial pressure of water in the atmosphere fell to 0.30 kPa. Will the frost sublime? What partial pressure of water would ensure...
-
On January 1, 2023, Bertrand, Incorporated, paid $86,900 for a 40 percent interest in Chestnut Corporation's common stock. This investee had assets with a book value of $229,500 and liabilities of...
-
Landen Corporation uses a job-order costing system. At the beginning of the year, the company made the following estimates: Direct labor-hours required to support estimated production Machine-hours...
-
Explain the concept of gene-environment interactions, and how these interactions influence the expression of complex traits and the risk of developing multifactorial diseases ?

Study smarter with the SolutionInn App


