Why is the magnitude of the electron affinity for a given element smaller than the magnitude of
Question:
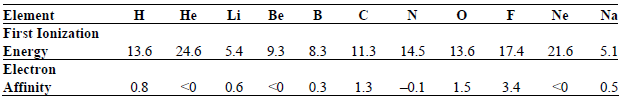
Transcribed Image Text:
в с Na Ne Не Li Be Element First Ionization н N 13.6 24.6 5.4 13.6 9.3 8.3 14.5 17.4 21.6 Energy Electron 11.3 5.1 0.3 <0 0.5 0.8 0.6 -0.1 3.4 1.5 Affinity <0 1.3 <0 3. 3. 3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
Within Koopmans approximation in which it is assumed that the electron distribution in an atom is un...View the full answer

Answered By

Jehal Shah
I believe everyone should try to be strong at logic and have good reading habit. Because If you possess these two skills, no matter what difficult situation is, you will definitely find a perfect solution out of it. While logical ability gives you to understand complex problems and concepts quite easily, reading habit gives you an open mind and holistic approach to see much bigger picture.
So guys, I always try to explain any concept keeping these two points in my mind. So that you will never forget any more importantly get bored.
Last but not the least, I am finance enthusiast. Big fan of Warren buffet for long term focus investing approach. On the same side derivatives is the segment I possess expertise.
If you have any finacne related doubt, do reach me out.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Explain why the first ionization energy and electron affinity for F are larger than for O.
-
The first ionization energy and electron affinity of Ar are both positive values. (a) What is the significance of the positive value in each case? (b) What are the units of electron affinity?
-
Use electron configurations to explain the following observations: (a) The first ionization energy of phosphorus is greater than that of sulfur. (b) The electron affinity of nitrogen is lower (less...
-
discuss the benefits and challenges of the employee job performance evaluation process as it relates to the ratings of an individual. For example, if an employer uses a standard Likert Scale (1...
-
In Exercises 59 and 60, determine whether the statement is true or false. Justify your answer. 1. The Leaning Tower of Pisa is not vertical, but when you know the angle of elevation to the top of...
-
The state of Michigan has just changed one of its toll roads from human collection of tolls to machine collection. The idea behind the change was to allow traffic to flow more smoothly through the...
-
The three bars are made of A-36 steel and form a pin-connected truss. If the truss is constructed when T 1 = 50F, determine the force in each bar when T 2 = 110F. Each bar has a cross-sectional area...
-
Consider customer lifetime value (CLV). Choose a business and show how you would go about developing a quantitative formulation that captures the concept. How would that business change if it totally...
-
Now consider the so-called Wagner-Fischer algorithm for computing the Levenshtein distance: WF (a[1...m], b[1...n]) Let D[0...m][0...n] be matrix of zeros For i in [1...m] do D[i][0] = i End For For...
-
The variable smokes is a binary variable equal to one if a person smokes, and zero otherwise. Using the data in SMOKE.RAW, we estimate a linear probability model for smokes: The variable white equals...
-
The electron affinities of He, Be, and Ne are negative, meaning that the negative ion is less stable than the neutral atom. Explain why this is so for these three elements.
-
Calculate the position of the maximum in the radial distribution function for Li 2+ in its ground state using the wave function in P21.13.
-
Let Z be a standard normal random variable. Use Appendix Table A.1 to find the numerical value for each of the following probabilities. Show each of your answers as an area under fZ(z). (a) P(0 Z ...
-
A block of inertia \(m\) is placed on an inclined plane that makes an angle \(\theta\) with the horizontal. The block is given a shove directly up the plane so that it has initial speed \(v\), and...
-
Identify the two most common responses of a defendant to an action for divorce and indicate the differences between them.
-
Identify the kinds of individuals who have standing to bring annulment actions.
-
Use the periodogram algorithm to estimate the PSD of \(L=1024\) samples of the following signal: \[x(n)=\cos \frac{2 \pi}{20} n+x_{1}(n)\] where \[x_{1}(n)=-0.9 x_{1}(n-1)+x_{2}(n),\] where...
-
Patel Company has an inexperienced accountant. During the first 2 weeks on the job, the accountant made the following errors in journalizing transactions. All entries were posted as made. 1. A...
-
You are looking to purchase a Tesla Model X sport utility vehicle. The price of the vehicle is $93,500. You negotiate a six-year loan, with no money down and no monthly payments during the first...
-
Banner Company acquires an 80% interest in Roller Company for $640,000 cash on January 1, 2013. The NCI has a fair value of $160,000. Any excess of cost over book value is attributed to goodwill. To...
-
The osmotic pressure of solutions of polystyrene in toluene were measured at 25C and the pressure was expressed in terms of the height of the solvent of density 1.004 g cm 3 : Calculate the molar...
-
A water carbonating plant is available for use in the home and operates by providing carbon dioxide at 5.0 atm. Estimate the molar concentration of the soda water it produces.
-
What proportions of hexane and heptane should be mixed (a) By mole fraction, (b) By mass in order to achieve the greatest entropy of mixing?
-
List 3 ways to organize a resume and describe when each resume is appropriate
-
Consider the following data for two products of Vigano Manufacturing. Activity Machine setup Parts handling Quality inspections Total budgeted overhead Unit Information Units produced Budgeted Cost...
-
ces Direct Materials Conversion Percent Percent Units Complete Complete Beginning work in process 42,500 inventory Units started this period 165,000 Units completed and 175,000 transferred out 32,500...

Study smarter with the SolutionInn App


