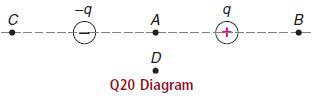
In the drawing for question 20, which point, B or C, will have the higher electric potential?
Question:
In the drawing for question 20, which point, B or C, will have the higher electric potential? Explain.
Transcribed Image Text:
-q A B D Q20 Diagram
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
In the above figure the field strengths at B and ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Physics of Everyday Phenomena A conceptual Introduction to physics
ISBN: 978-0073512112
6th edition
Authors: W. Thomas Griffith, Juliet W. Brosing
Question Posted:
Students also viewed these Physics questions
-
Will a negatively charged particle, initially at rest in an electric field, tend to move toward a region of lower electric potential if released? Explain.
-
Two charged particles exert an electrostatic force of 8 N on each other. What will the magnitude of the electrostatic force be if the distance between the two charges is reduced to one-half of the...
-
A +1.5 x 10 -6 C test charge experiences forces from two other nearby charges: a 12-N force due east and an 8-N force due west. What are the magnitude and direction of the electric field at the...
-
In Exercises 1 through 14, compute the indicated values of the given function. f(x) = 3x 2 + 5x 2; f(0), f(2), f(1)
-
On April 3, 2013, Terry purchased and placed in service a building. The building cost $2 million. An appraisal determined that 25% of the total cost was attributed to the value of the land. The...
-
What possible geometrical forms can the nodes in the angular function for p and d orbitals in the H atom have? What possible geometrical forms can the nodes in the radial function for s, p, and d...
-
The change in entropy of an ideal gas during a reversible isothermal expansion is (a) Zero (b) Positive (c) Negative (d) Infinite.
-
Howski Associates is an independent insurance agency that sells business, automobile, home, and life insurance. Maya Howski, senior partner of the agency, recently attended a workshop at the local...
-
America's relationship with its mentally ill population continues to suffer due to inadequacies in the country's mental health care system. For the mentally ill in Chicago, the effects of this...
-
To study the growth of pine trees at an early stage, a nursery worker records 40 measurements of the heights (cm) of one-year-old red pine seedlings, (courtesy of Professor Alan Ek) The summary...
-
Is electric potential the same as electric potential energy? Explain.
-
Would you be more likely to be struck by lightning if you stood on a platform made from a good electrical insulator than if you stood on the ground? Explain.
-
For flow in a chip with square channels 300 m on a side, what fluid velocity, in m/s, corresponds to a Reynolds number of 10, if the fluid is water at 20C? What if it is air at 70C?
-
A student volunteers to gather information on a company for a group case analysis project. The other members of the group agree and tell her to go ahead and choose the information sources. In terms...
-
The ____________ structure tries to combine the best elements of the functional and divisional forms. (a) virtual (b) boundary less (c) team (d) matrix
-
Rumours and resistance to change are potential disadvantages often associated with ____________. (a) virtual organizations (b) informal structures (c) delegation (d) specialized staff
-
True internalization and commitment to a planned change is most likely to occur when a manager uses a(n)_________ change strategy. (a) education and communication (b) rational persuasion (c)...
-
The control equation states:________ = Desired Performance Actual Performance. (a) Problem Magnitude (b) Management Opportunity (c) Planning Objective (d) Need for Action
-
In January 2014, the management of Stefan Company concludes that it has sufficient cash to permit some short-term investments in debt and share securities. During the year, the following transactions...
-
Match each of the key terms with the definition that best fits it. _______________ A record of the sequence of data entries and the date of those entries. Here are the key terms from the chapter. The...
-
A symmetric dielectric slab waveguide has a slab thickness d = 10m, with n 1 = 1.48 and n 2 = 1.45. If the operating wavelength is = 1.3m, what modes will propagate?
-
Assume a raised-cosine pulse with β = 0.2 additive noise with power spectral density and a channel filter with transfer-function-squared magnitude given by Find and plot the optimum...
-
(a) Sketch the trapezoidal spectrum (b) By appropriate sketches, show that it satisfies Nyquists pulse-shaping criterion. P(f) =A(f /b) A(F/a), b > a > 0, for a =1 and b = 2. %| b-a b-a
-
Assume you are a trader with JP Morgan. From the quote screen on your computer terminal, you notice that Bank A is quoting 0.8354/$1.00 and Bank Bis offering SF1.0913/$1.00. You learn that Bank Cis...
-
Leven Clinic uses client-visits as its measure of activity. During September, the clinic budgeted for 3,000 client-visits, but its actual level of activity was 3,050 client-visits. The clinic has...
-
If 2.25 grams of calcium hydroxide is dissolved in 154.56 ml of water, a. What's the Molarity (M), if the volume of solution is 500.00 ml? b. What's the Molarity (M) of hydroxide ions, if the volume...

Study smarter with the SolutionInn App


