0.10 mol of nitrogen gas follow the two processes shown in Figure P19.58. How much heat is...
Question:
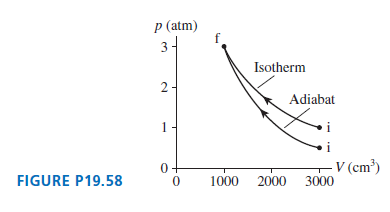
Transcribed Image Text:
p (atm) 3 Isotherm 2- Adiabat V (cm³) 3000 FIGURE P19.58 1000 2000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Model We have an adiabatic and an isothermal process Solve For the adiabatic process no heat is add...View the full answer

Answered By

Rustia Melrod
I am a retired teacher with 6 years of experience teaching various science subjects to high school students and undergraduate students. This background enables me to be able to help tutor students who are struggling with the science of business component of their education. Teaching difficult subjects has definitely taught me patience. There is no greater joy for me than to patiently guide a student to the correct answer. When a student has that "aha!" moment, all my efforts are worth it.
The Common Core standards are a useful yardstick for measuring how well students are doing. My students consistently met or exceeded the Common Core standards for science. I believe in working with each student's individual learning styles to help them understand the material. If students were struggling with a concept, I would figure out a different way to teach or apply that concept. I was voted Teacher of the Year six times in my career. I also won an award for Innovative Teaching Style at the 2011 National Teaching Conference.
4.90+
4+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
How much heat is required to change a 1 kg block of ice at 10oC to steam at 110oC? Give your answer in Joule and calorie and Calorie. (1 cal = 4.186 J; 1 Calorie = 1000 calorie.
-
How much heat is required to melt a 2.5-kg block of ice at 0oC?
-
How much heat is required to boil away 1.50 kg of water that is initially at 100oC?
-
Attlee Ltd holds 28% of the issued shares of Nehru Ltd. Attlee Ltd acquired these shares on 1 July 2019 and on this date all the identifiable assets and liabilities of Nehru Ltd were recorded at...
-
Blue Company, an architectural firm, has a bookkeeper who maintains a cash receipts and disbursements journal. At the end of the year (2016), the company hires you to convert the cash receipts and...
-
Aspirations Ltd commenced trading as wholesale suppliers of office equipment on 1 January 20X1, issuing ordinary shares of £1 each at par in exchange for cash. The shares were fully paid on...
-
A turbine rotor is run at the natural frequency of the system. A stroboscope indicates that the maximum displacement of the rotor occurs at an angle \(229^{\circ}\) in the direction of rotation. At...
-
Debra Menge started her own consulting firm, Menge Consulting, on May 1, 2014. The following transactions occurred during the month of May. May 1 Debra invested $7,000 cash in the business. 2 Paid...
-
An instructor is performing Young's double-slit experiment for his students. He directs a beam of laser light to a pair of parallel slits, which are separated by 0.102mmfrom each other. The portion...
-
Jennifer Lopez opened a school for administrative skills called Lopez Office Training and completed the following transactions: a. Contributed the following assets to the business: Cash ...........
-
0.10 mol of nitrogen gas follow the two processes shown in Figure EX19.31. How much heat is required for each? p (atm) 3 2 - V (cm) 0+ 2000 3000 1000 FIGURE EX19.31
-
You come into lab one day and find a well-insulated 2000 mL thermos bottle containing 500 mL of boiling liquid nitrogen. The remainder of the thermos has nitrogen gas at a pressure of 1.0 atm. The...
-
Bond laddering is ________. a. A risky bond investment strategy that may yield tremendous returns b. A strategy in which bonds with several different maturity periods are added to a portfolio c. A...
-
Can necessity ever justify homicide? Why or why not?
-
Ten-year-old Esmeralda Guevara wrote a note on a stall in the girls bathroom at a school in College Place, Washington, on the afternoon of Monday, November 5, 2007. The note read, [B]omb set 20 mins...
-
Identify four types of intellectual property.
-
Compare and contrast larceny and embezzlement.
-
Why can a duress defense rarely succeed in homicide cases?
-
Find the equation of the line containing the points (-1, 4) and (2, -2). Express your answer in slope-intercept form and graph the line.
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
What do radio waves and light have in common? What is different about them?
-
When you look downward at a fish in water, does refraction make the fish appear closer to the surface or deeper?
-
When you view the Sun near the horizon, does refraction make it appear higher or lower in the sky?
-
Outdoor Luggage, Incorporated, makes high-end hard-sided luggage for sports equipment. Data concerning three of the company's most popular models appear below. Ski Guard Selling price per unit $ 270...
-
Jury nullification can be defined as when the jury refuses to convict a defendant who is legally guilty of the crime. This typically occurs when a jury finds that the law is unjust or that the...
-
21. (5 points) Write code snippet to find the smaller number of two integers. int num1 = input.nextInt(); int num2=input.nextInt(): int smaller; // your code here 22. (5 points) Write code snippet to...

Study smarter with the SolutionInn App


