8.0 g of helium gas follows the process 1 ?? 2 ?? 3 shown in FIGURE P18.60.
Question:
8.0 g of helium gas follows the process 1 ?? 2 ?? 3 shown in FIGURE P18.60. Find the values of V1, V3, p2, and T3.
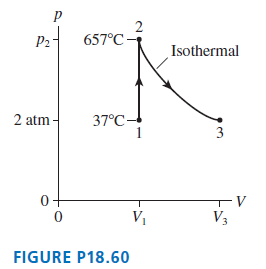
Transcribed Image Text:
P2- 657°C - Isothermal 2 atm - 37°C- 3 0+ -v V1 V3 FIGURE P18.60
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Model Assume that the helium gas is an ideal gas Visualize Process 1 2 is isochoric proc...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Consider the following gas samples: 4.0 g of hydrogen gas, 4.0 g of helium gas, 1.0 mole of fluorine gas, 44.0 g of carbon dioxide gas, and 146 g of sulfur hexafluoride gas. Arrange the gas samples...
-
A monatomic gas follows the process 1 2 3 shown in Figure EX19.32. How much heat is needed for (a) Process 1 2 (b) Process 2 3? p (atm) 3- 2- 100C isotherm 3 -V (cm) 300 100 200 FIGURE EX19.32
-
Consider a container like that shown in Figure 20.12, with n 1 moles of a monatomic gas on one side and n 2 moles of a diatomic gas on the other. The monatomic gas has initial temperature T 1i ....
-
Court Casuals has 100,000 shares of common stock outstanding as of the beginning of the year and has the following transactions affecting stockholders' equity during the year. May 18 Issues 25,000...
-
Mauve, Inc., uses a two- to six-year graded vesting approach in its retirement plan. Calculate the nonforfeitable percentage for each of the following participants based upon the years of service...
-
Finagle A Bagel, a fast-growing New England small business co-owned by Alan Litchman and Laura Trust, is at the forefront of one of the freshest concepts in the food-service business: fresh food. The...
-
A diesel engine of mass \(300 \mathrm{~kg}\) and operating speed \(1800 \mathrm{rpm}\) is found to have a rotating unbalance of \(1 \mathrm{~kg}-\mathrm{m}\). It is to be installed on the floor of an...
-
Rehab Health Care, LLC, incurred the following service-related activity costs for the month: Total sales ................$42,000 Customer complaint processing ....... 1,200 Employee training...
-
Robert and Sandra have two children, not disabled, ( ages , 3 and 4 year old ) This year, Robert\'s Earned Income is $ 3 0 , 0 0 0 and Sandra\'s is $ 6 0 , 0 0 0 . They paid a Nanny $ 5 , 0 0 0 this...
-
Read the given description of a CSP and write solution as per the given questions: [20] YCPS@GIFT is going to arrange a gaming competition at campus. They are considering using seven games for this...
-
Texas Instruments (TI) designs and manufactures semiconductor products for use in computers, telecommunications equipment, automobiles, and other electronics-based products. The manufacturing of...
-
JCPenney operates a chain of retail department stores, selling apparel, shoes, jewelry, and home furnishings. It also offers most of its products through catalog distribution. During fiscal Year 5,...
-
How would the shopping behaviors of those on each end of the ethnocentrism continuum differ?
-
Take the procedural design approach and create first the context diagram, and then the highest - level data flow and control - flow diagrams for an electronic lock in the laboratory door having the...
-
Suppose that, in order to protect Ronaldo from his adoring fans, soccer teams that host Real Madrid must hire extra security, and security costs go up as the number of fans at the game goes up. When...
-
N.B. Vargaftik 3 (1975) lists the experimental values in the following table for the enthalpy departure of isobutane at 175C. Compute theoretical values and their percent deviations from experiment...
-
Suppose the can segment their fans into young fans and senior citizens. Young fans have the demand curve 120 10G (MR = 120 20G). Senior citizens have the demand curve p = 60 10G (MR = 60 20G)....
-
Recent research in thermodynamic perturbation theory suggests the following equation of state. (a) Derive the departure function for (A A ig ) T,V . (b) Derive the departure function for (U U ig )....
-
Describe the main requirements of the Sarbanes-Oxley Act of 2002.
-
Consider the advantages and disadvantages of extending property rights so that everyone would have the right to prevent people imposing any costs on them whatsoever (or charging them to do so).
-
In which of the four basic interactions does an electron participate? A neutrino? A proton? A photon?
-
What is a quark? How many different types of quarks are now known? What are some of the basic properties that distinguish these quarks?
-
Describe the kinds of evidence that have led scientists to conclude that quarks exist.
-
Energy market contains three generation companies and three consumers. The energy bids and offers submitted are presented in the following tables. Plot the supplies and consumer curves. Determine the...
-
During a three - year period, Appliances, Inc., performed electrical heating and plumbing work for Yost Construction worth approximately $ 7 , 0 0 0 . Yost never paid Appliances for any of these...
-
ABC and BCD Joint Corporation is a division of a major corporation. Last year the division had total sales of SAR 8 5 , 7 8 0 , 0 0 0 , net operating income of SAR 8 , 6 9 7 , 5 7 0 , and average...

Study smarter with the SolutionInn App


