Draw a series of pictures, similar to Figure 41.21, for the ground states of Ca, Ni, As,
Question:
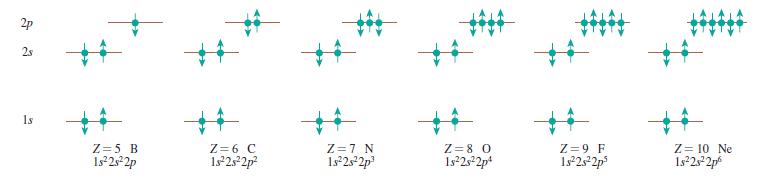
Draw a series of pictures, similar to Figure 41.21, for the ground states of Ca, Ni, As, and Kr.
Figure 41.21:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
For the ground state ...View the full answer

Answered By

Sheeba Khan
I have been a chemistry teacher since 2014 and started my career as an online tutor in 2018. I love to study and teach chemistry. It is my passion.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Draw a series of pictures, similar to Figure 41.22, for the ground states of Ca, Ni, As, and Kr. 2p 2s %23 %23 1s Z = 5 B 1s25 2p Z = 6 C 1s 2s2p? Z =7 N 1s 2s2p Z = 8 0 1s2s2p* Z = 9 F 1s 2s2p Z =...
-
Draw a series of pictures, similar to Figure 41.22, for the ground states of K, Sc, Co, and Ge. 2p 2s %23 %23 1s Z = 5 B 1s25 2p Z = 6 C 1s 2s2p? Z =7 N 1s 2s2p Z = 8 0 1s2s2p* Z = 9 F 1s 2s2p Z = 10...
-
Draw a series of pictures, similar to Figure 41.21, for the ground states of K, Sc, Co, and Ge. Figure 41.21:
-
If you can't find similar ratios to class-covered ones, use financial formulas to calculate them manually. Choose up to two of the following: Operating Margin EBITDA Margin Payout Ratio 3. Analysis...
-
A variable of two populations has a mean of 40 and a standard deviation of 12 for one of the populations and a mean of 40 and a standard deviation of 6 for the other population. a. For independent...
-
Youve conducted a study to determine how the number of calories a person consumes in a day correlates with time spent in vigorous bicycling. Your sample consisted of ten women cyclists, all of...
-
Much like the medical and legal fields, members of the actuarial profession face interesting problems and are generally well compensated for their efforts in resolving these problems. Also like the...
-
Three methods of interperiod income tax allocation have been advocated. These include (1) the asset/liability method, (2) the deferred method, and (3) the net-of-tax method. Required Define...
-
Write a Java application that will request a user to enter a sentence, after which it will perform the following operations on the sentence: . Firstly, it will split the sentence into individual...
-
On October 1, 2019, Helen Kennedy opened an advertising agency. She plans to use the chart of accounts listed below. INSTRUCTIONS 1. Journalize the transactions. Number the journal page 1, write the...
-
Figure 41.22 shows that the ionization energy of cadmium (Z = 48) is larger than that of its neighbors. Why is this? Figure 41.22:
-
a. Draw a diagram similar to Figure 41.2 to show all the possible orientations of the angular momentum vector L for the case l = 3. Label each L with the appropriate value of m. b. What is the...
-
During 2016, Schottenheim Corporation buys laptop computers and desktop computers to use in its general sales offices. Schottenheim buys laptops for $42,000 on March 29, additional laptops for...
-
How are weekends and holidays handled when volatility is estimated and used?
-
In the Black-Scholes-Merton model, what happens to the standard deviation of the continuously compounded rate of return as the length of the time period over which it is measured increases?
-
Explain the two main ways that derivatives trade?
-
What are the properties of a Wiener process?
-
Explain the role of the Hurst exponent in fractional Brownian motion.
-
An electrode is prepared from liquid mercury in contact with a saturated solution of mercury(I) chloride, Hg2Cl2, containing 1.00 M Cl. The cell potential of the voltaic cell constructed by...
-
Consider the reaction of acetic acid in water CH 3 CO 2 H(aq) + H 2 O(l) CH3CO 22 (aq) + H 3 O + (aq) where Ka 5 1.8 3 1025. a. Which two bases are competing for the proton? b. Which is the stronger...
-
Which of the following transitions in sodium do not occur as electric dipole transitions? (Give the selection rule that is violated.) 451/2 3S1/2 4D3/23P1/2 4S1/2 3P3/2 4D3/2 351/2 4P3/2 3S1/2 5D3/2...
-
Write the ground-state electron configuration of (a) Carbon, (b) Oxygen, and (c) Argon.
-
Five identical noninteracting particles are place in an infinite square well with L = 1.0 nm. Compare the lowest total energy for the system if the particles are (a) Electrons and (b) Pions. Pions...
-
Example 1: Water flows through a 200-mm diameter watermain. Over a 500-m length, the hydraulic gradient line falls 10 m. The absolute roughness of the watermain is 0.20 mm. The kinematic viscosity of...
-
Given: A 1-kg, AISI 1070 high-carbon steel is being cooled slowly to the following temperatures (a) 800 C; (b) 728 C; and (c) 726 C Find: Determine the amount of gamma, alpha and cementite phases in...
-
Use the Chain Rule to find Oz/as and az/at. z=x6y8, x = s cost, y = s sin t Oz = as Oz Ot =

Study smarter with the SolutionInn App


