FIGURE EX20.10 is a histogram showing the speeds of the molecules in a very small gas. What
Question:
FIGURE EX20.10 is a histogram showing the speeds of the molecules in a very small gas. What are
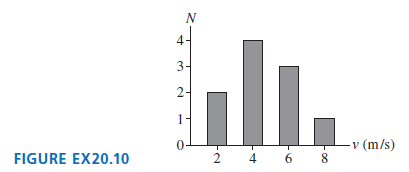
(a) The most probable speed,
(b) The average speed,
(c) The rms speed?
Transcribed Image Text:
N In. 3- 2- -v (m/s) FIGURE EX20.10 2 4 6 8 4.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (15 reviews)
Solve a The most probable s...View the full answer

Answered By

Ajeet Singh
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students. Areas of interest: Business, accounting, Project management, sociology, technology, computers, English, linguistics, media, philosophy, political science, statistics, data science, Excel, psychology, art, history, health education, gender studies, cultural studies, ethics, religion. I am also decent with math(s) & Programming. If you have a project you think I can take on, please feel welcome to invite me, and I'm going to check it out!
5.00+
4+ Reviews
24+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
As a sample of nitrogen gas (N2) undergoes a temperature increase at constant volume, the distribution of molecular speeds increases. That is, the probability distribution function P(v) for the...
-
(a) Place the following gases in order of increasing average molecular speed at 300 K: CO, SF6, H2S, Cl2, HBr. (b) Calculate and compare the rms speeds of CO and Cl2 molecules at 300 K. (c) Calculate...
-
A molecular beam apparatus employs supersonic jets that allow gas molecules to expand from a gas reservoir held at a specific temperature and pressure into a vacuum through a small orifice. Expansion...
-
2. A magazine printer is considering taking on a new weekly publication. The company's financial officer has researched and determined costs and a committee of upper management personnel are deciding...
-
Paola and Isidora are married; file a joint tax return; report modified AGI of $148,000; and have one dependent child, Dante. The couple paid $12,000 of tuition and $10,000 for room and board for...
-
BigCos chief financial officer is trying to determine a fair value for PrivCo, a nonpublicly traded firm that BigCo is considering acquiring. Several of PrivCos competitors, Ion International and...
-
Starting from fundamentals, show that the equation for the lateral vibration of a circular membrane is given by \[\frac{\partial^{2} w}{\partial r^{2}}+\frac{1}{r} \frac{\partial w}{\partial...
-
What should Mr. Rutland do? Include the following issues: Budget: acquiring adequate resources Philanthropic organizations Governmental agencies Donations Memberships Visitors Budget:...
-
Suppose soda was 2 dollars a can, and pop was 1 dollar a can. The total amount of money that Frank has is 10 Dollars. Draw a graph with budget constraints. Please DRAW the indifference curve. How...
-
How might sleep deprivation influence aspects of expectancy theory? How might the incorporation of nap rooms for sleep-deprived employees influence aspects of equity theory?
-
Eleven molecules have speeds 15, 16, 17, , 25 m/s. Calculate (a) v avg (b) v rms .
-
The number density in a container of neon gas is 5.00 10 25 m -3 . The atoms are moving with an rms speed of 660 m/s. What are (a) The temperature (b) The pressure inside the container?
-
You are tracking melanoma in your county. Calculate the race- and gender-specific mortality rates from the data provided in Table 2-10. Men Women White African American New Cases 5 3 7 1 Deaths 2 1 3...
-
Which of the following is a good example of microeconomics? a. How a specific company would maximize its production and capacity so it could better compete in its industry b. How an increase in the...
-
Ethics is the study of the general nature of morals and the specific moral choices a person makes. True or False
-
Racerback Swimwear, an Australian company, opens a factory near Tallahassee, Florida. The value of the swimsuits produced in the new Florida factory is included in which countrys GDP? a. Australia b....
-
Minimizing a companys carbon footprint is part of a plan to increase corporate philanthropy. True or False
-
Which of the following is an example of the governments fiscal policy to stimulate an economy? a. Increased government spending b. Increased taxes c. Decreases in the discount rate d. Selling...
-
What is the Microsoft.NET Framework? What basic elements does it include?
-
As long as we can't lose any money, we have a risk-free investment." Discuss this comment. Q2: Both investing and gambling can be defined as "undertaking risk in order to earn a profit." Explain how...
-
What kind of nuclear power is responsible for sunshine?
-
Throw ten coins onto a flat surface. Move aside all the coins that landed tails-up. Collect the remaining coins. After tossing them once again, remove all coins that landed tails-up. Repeat this...
-
Repeat the above, but use 10 dimes and 25 pennies. Let the dimes represent a radioactive isotope, such as carbon-14, while the pennies represent a nonradioactive isotope, such as carbon-12. Remove...
-
what are some ways to build a great organizational culture so that people are content and productive in their work? How would you define a great organizational culture?
-
On December 3 1 , 2 0 2 3 , Cheyenne Ltd . has $ 3 1 4 3 0 0 0 in short - term notes payable due on February 1 4 , 2 0 2 4 . On January 1 0 , 2 0 2 4 , Cheyenne arranged a line of credit with Caldi...
-
The following information pertains to Mason Company for Year 2 . Beginning inventory 1 1 6 units @ $ 3 0 Units purchased 3 5 8 units @ $ 4 5 Ending inventory consisted of 4 2 units. Mason sold 4 3 2...

Study smarter with the SolutionInn App


