The heat engine shown in FIGURE P21.63 uses 0.020 mol of a diatomic gas as the working
Question:
The heat engine shown in FIGURE P21.63 uses 0.020 mol of a diatomic gas as the working substance.
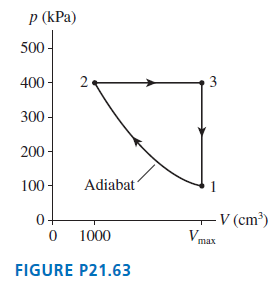 a. Determine T1, T2, and T3.b. Make a table that shows ΔEth, Ws, and Q for each of the three processes.c. What is the engine??s thermal efficiency?
a. Determine T1, T2, and T3.b. Make a table that shows ΔEth, Ws, and Q for each of the three processes.c. What is the engine??s thermal efficiency?
Transcribed Image Text:
Р (КРа) 500 - 400 - 2. 3 300 - 200 - 100- Adiabat – (cm³) Vmax 1000 FIGURE P21.63
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Model The closed cycle in this heat engine includes adiabatic process 1 2 ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
The heat engine shown in FIGURE P21.62 uses 2.0 mol of a monatomic gas as the working substance. a. Determine T 1 , T 2 , and T 3 . b. Make a table that shows ÎE th , W s , and Q for each of...
-
A heat engine using a monatomic gas follows the cycle shown in FIGURE P21.58. a. Find W s , Q, and E th for each process in the cycle. Display your results in a table.b. What is the thermal...
-
(a) Calculate the total rotational kinetic energy of the molecules in 1.00 mol of a diatomic gas at 300 K. (b) Calculate the moment of inertia of an oxygen molecule (O2) for rotation about either the...
-
Adrian Webb bought 618 bricks to build a barbecue pit, paying $185.40. Find the cost per brick. (HINT: Cost per brick means the cost for one brick.) The cost per brick is s (Round to nearest hundidth)
-
Who in a firm should be responsible for industry analysis? Assume that the firm does not have a strategic planning department.
-
Without using a calculator, indicate be-tween which two adjacent ruler marks each of the following irrational numbers will fall. Support your answer by obtaining an approximation with a calculator....
-
If a material expands freely due to heating, it will develop (a) Tensile stress (b) Compressive stress (c) Shear stress (d) No stress
-
The three following diagrams show the supply of luxury ocean liners at three different levels of aggregationthe entire world, a particular country, and a particular firm. a. Which diagram shows the...
-
What is the present value of 10 payments of $500 each received every 24 months at a discount rate of 12% compounded annually?
-
Find the main dimension of 100 MVA, 11 kV, 50 HZ, 150 rpm, three-phase water wheel generator. The average gap density = 0.65 Wb/m2 and ampere conductors/m are 40000. The peripheral speed should not...
-
A heat engine using 120 mg of helium as the working substance follows the cycle shown in FIGURE P21.61. a. Determine the pressure, temperature, and volume of the gas at points 1, 2, and 3.b. What is...
-
A heat engine with 0.20 mol of a monatomic ideal gas initially fills a 2000 cm 3 cylinder at 600 K. The gas goes through the following closed cycle: Isothermal expansion to 4000 cm 3 . Isochoric...
-
Protectionist trade policies are often enacted with the political intention of strengthening exports, and hence GDP. Examples of protectionist trade policies include quotas and import taxes. What is...
-
Lindal Corporation, organized in 2023, immediately filed an election for S corporation status under the rules of Subchapter S. What is the maximum amount of passive investment income that Lindal will...
-
Use the following information to answer questions 28-33: The following is the list of activities and the crashing cost for each task. Activity Normal time (weeks) K L N M OPYN Z End Immediate...
-
= Let Q {q} be an orthonormal basis for a subspace V of R". a) Prove that if vV and v-q=0 for all i then v = 0. [2] 1 b) Prove that if v V then v = (v qq (Hint. Take the difference and evaluate the...
-
On January 1, 2024, the general ledger of Dynamite Fireworks includes the following account balances: Accounts Cash Debit $24,900 Credit Accounts Receivable 6,300 Supplies 4,200 Land 61,000 Accounts...
-
In this exercise, let's pretend there's only one make, model and age of used cars. A used car can either be high quality or low quality. It is known that half of all existing used cars are high...
-
What MAC protocol is used in the baseband layer of Bluetooth?
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
What is the ratio of the Earths orbital period about the sun to the Earths period of rotation about its own axis?
-
Joe has a weight of 720 N (about 162 lb) when he is standing on the surface of the Earth. What would his weight (the gravitational force due to the Earth) be if he doubled his distance from the...
-
Two masses are attracted by a gravitational force of 0.36 N. What will the force of attraction be if the distance between the two masses is tripled?
-
For each ordered pair, determine -6x+7y=2 7x-4y=6 a solution to the system of equations.
-
Review the meaning of the concepts or terms given in Key Terms and Concepts. Distinguish between a production department and a service department. Distinguish between a direct cost and an indirect...
-
What would you like the members of your learning community to know about your global health interests, perspectives, and life experiences? How will you prepare to listen and learn from others? Why...

Study smarter with the SolutionInn App


