Use the data from Figure 40.24 to calculate the first three vibrational energy levels of a C
Question:
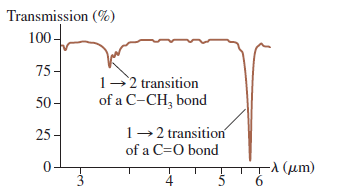
Transcribed Image Text:
Transmission (%) 100- 75- 1-2 transition of a C-CH, bond 50- 1-2 transition 25- of a C=0 bond -λ (μm) 6.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Solve From Figure 4024 we see that absorption due to the C O bond is at 5...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
For IF ( = 610 cm 1 ) calculate the vibrational partition function and populations in the first three vibrational energy levels for T = 300 and 3000 K. Repeat this calculation for IBr ( = 269 cm 1 )....
-
The first five vibrational energy levels of HI are at 1144.83, 3374.90, 5525.51, 7596.66, and 9588.35 cm-1 Calculate the dissociation energy of the molecule in reciprocal centimeters and electron...
-
The vibration frequency for the molecule HF is 1.24 x 1014 Hz. The mass of a hydrogen atom is 1.67 x 10-27 kg, and the mass of a fluorine atom is 3.15 x 10-26 kg. (a) What is the force constant k'...
-
Exporting files into binary files is done in O Logical files O Physical backup O Physical full backup O Physical partial backup L
-
What is the main difference between speed and velocity?
-
Dover Chemical Company manufactures specialty chemicals by a series of three processes, all materials being introduced in the Distilling Department. From the Distilling Department, the materials pass...
-
Nicole Lutes, a partner in a regional public accounting firm, is conducting the independent technical reviewthe "cold" reviewof working papers for the December 31, 1999, year end audit of Singer...
-
Ichiro Manufacturing Co. uses the process cost system. The following information for the month of December was obtained from the company's books and from the production reports submitted by the...
-
Find the minimal equivalent DFA. Explanation on how you did the work 10 a b a, b 91 92 93 94 b a, b b a, b 95
-
Kris Allen Company established a petty cash fund on May 1, cashing a cheque for $150. The company reimbursed the fund on June 1 and July 1 with the following results. On July 10, Kris Allen increased...
-
An electron is confined in a harmonic potential well that has a spring constant of 12.0 N/m. What is the longest wavelength of light that the electron can absorb?
-
Verify that the n = 1 wave function 1 (x) of the quantum harmonic oscillator really is a solution of the Schrdinger equation. That is, show that the right and left sides of the Schrdinger equation...
-
Draw a normal curve with = 50 and = 5. Label the mean and the inection points.
-
The data in the following table were obtained in a travel time study on a section of highway with length 1.5 km, using the moving-vehicle technique. Determine average travel time, volume, and speed...
-
Water flows through the joint as shown below. Find the horizontal and vertical components of the force acting on the joint because of the flow of water. Neglect energy loss and body forces. A = 0.1 m...
-
Problem 11 A bullet of mass 0.0020 kg embeds itself in a wooden block with mass 0.991 kg, which then compresses a spring (k = 190 N/m) by a distance 0.055 m before coming to rest. The coefficient of...
-
5. A mass on a vertical spring is observed to have the displacement vs. time graph below. From the graph, what is the period of the motion in seconds? Explain or show how you got your answer. 6. If...
-
high service level application requires 500 tons of salt per inch of snow, which would require 11,000 tons of salt, if 2 inches of snow fell. These numbers are consolidated at Exhibit 3. Sarah...
-
A fully amortizing mortgage loan is made for $100,000 at 6 percent interest for 20 years. a. Calculate the monthly payment for a CPM loan. b. What will the total of payments be for the entire 20-year...
-
What is EBIT/eps analysis? What information does it provide managers?
-
Suppose that you drop BBs onto a bulls-eye marked on the floor. According to the uncertainty principle, the BBs do not necessarily fall straight down from the release point to the center of the...
-
It is possible for some fundamental particles to violate conservation of energy by creating and quickly re-absorbing another particle. For example, a proton can emit a + according to p = n + + ,...
-
An electron and a positron are moving toward each other with equal speeds of 3 x 10 6 m/s. The two particles annihilate each other and produce two photons of equal energy. (a) What were the de...
-
2-Using the CPT data in figure below, a unit weight of 115 pcf, and an overconsolidation ratio of 3, determine (a) soil behavior type (b) o' using Kulhawy and Mayne correlation and (c) N60 for the...
-
How does the application of computational fluid dynamics (CFD) coupled with multiphase flow modeling enhance the understanding and prediction of complex hydrodynamic behaviors in industrial-scale...
-
1. From the joint distribution defined by the graphical model, determine if the conditional independence holds: ALL FIE, B, and provide brief explanation (5 points). 2. Given an undirected chain...

Study smarter with the SolutionInn App


