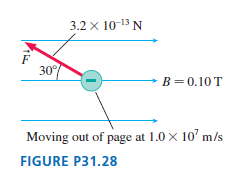
What are the electric field strength and direction at the position of the electron in FIGURE P31.28
Question:
What are the electric field strength and direction at the position of the electron in FIGURE P31.28 ?
Transcribed Image Text:
3.2 x 10-13 N F 30° B=0.10 T Moving out of page at 1.0 x 10' m/s FIGURE P31.28
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
Model Assume the electric and magnetic fields are uniform Solv...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
What electric field strength and direction will allow the proton in FIGURE P31.30 to pass through this region of space without being deflected? B= 0.010 T- v = 2.0 x 107 m/s FIGURE P31.30 +
-
What are the magnetic field strength and direction at points a to c in FIGURE EX29.14? a. ]2.0 cm 10 A b. 4.0 cm 10 A [2.0 cm c. FIGURE EX29.14
-
What magnetic field strength and direction will levitate the 2.0 g wire in FIGURE EX29.33? 2.0 g wire B-field region 1.5 A 10 cm FIGURE EX29.33
-
Generally, the JDBC API perform the following three functions a. Connect to database, load JDBC driver, perform the query b. Perform the query, connect to database, load JDBC driver c. Get result...
-
A large punch bowl holds 3.99 kg of lemonade (which is essentially water) at 20.5 C. A 0.0550-kg ice cube at -10.2 C is placed in the lemonade. What are the final temperature of the system, and the...
-
Propyl alcohol (CH3CH2CH2OH) and isopropyl alcohol [(CH3)2CHOH], whose space-filling models are shown, have boiling points of 97.2 oC and 82.5 oC, respectively. Explain why the boiling point of...
-
Consider the multiple regression model fit to the house price data in Problem 3.7. Problem 3.7 Consider the house price data in Table B.4. a. Construct a normal probability plot of the residuals....
-
Napa Tours Co. is a travel agency. The nine transactions recorded by Napa Tours during April 2018, its first month of operations, are indicated in the following T accounts: Indicate for each debit...
-
How do organizational learning theories, such as the learning organization concept and knowledge management practices, contribute to sustained competitive advantage and organizational resilience in...
-
Florida Power & Light (FP&L) Company has enjoyed a reputation for quickly fixing its electric system after storms. However, during the hurricane seasons of 2004 and 2005, a new reality was that the...
-
A 200 mW vertically polarized laser beam passes through a polarizing filter whose axis is 35 from horizontal. What is the power of the laser beam as it emerges from the filter?
-
What is the force (magnitude and direction) on the proton in FIGURE P31.29? Give the direction as an angle cw or ccw from vertical. E= 1.0 x 10 V/m x / x v = 1.0 x 10' m/s B= 0.10 T FIGURE P31.29
-
By calculating the standard emf, decide whether aqueous sodium hypochlorite solution will oxidize Fe2+ (aq) to Fe3+ (aq) in basic solution under standard conditions. See Appendix I for data.
-
The rate of hydrolysis, K, of methyl acetate is 4 times faster at 25 C than at 4.6 C. Using the Arrhenius expression for reaction rate, Ea K = K exp(-4) KBT where Ko, the theoretical rate of...
-
In the protein, the side chains of the amino acids (denoted by R) are exposed to different environments in the cell. Below is a fragment of a cell membrane with the channel protein spanning the...
-
1. A student determines the freezing point of a solution of 0.92 g of unknown in 23.78 g of t-butyl alcohol. He obtains the following time and temperature readings (times are in minutes, temperatures...
-
A student proposes the following Lewis structure for the dinitrogen monoxide (NO) molecule. N=0=N Assign a formal charge to each atom in the student's Lewis structure. atom left N ** O right N formal...
-
Describe the separation techniques of decantation, filtration, extraction, and sublimation. Compare homogenous and heterogenous mixtures, provide a definition for each one of them and state the...
-
In the basic EOQ model in Example 12.1, suppose that the fixed cost of ordering is $500. Use Solver to find the new optimal order quantity. How does it compare to the optimal order quantity in the...
-
Sandcastles, Inc.s management has recently been looking at a proposal to purchase a new brick molding machine. With the new machine, the company would not have to buy bricks. The estimated useful...
-
The existence of the anti-hyperons was proven by the discovery of an anti-lambda by M. Baldo-Ceolin and D. J. Prowse in 1958. A beam of negative pions with energy E = 4.6 GeV hit an emulsion stack....
-
For each of the following reactions (a) establish whether it is allowed or not, (b) if it is not, give the reasons (they may be more than one), (c) give the types of interaction that allow it: (1) -...
-
For each of the following reactions establish whether it is allowed or not; if it is not, give the reasons: (1) + e + + ; (2) e e + ; (3) p + p + K + ; (4) p+p p + + + K - ; (5) p e + + e ;...
-
Create a segmentation, selection and focus model that allows the PUMA company in Mexico to determine its customer, where to find it and how to generate value given its model. 7) Selection of the most...
-
Determine if the following values are zeroes polynomial. 4 3 f(x) = x + x-18x - 16x + 32 x+1, +2, +4 -
-
Cali Windows is a small company that installs windows. Its cost structure is as follows: (Click the icon to view the cost structure.) Calculate (a) the breakeven point in units and revenues and (b)...

Study smarter with the SolutionInn App


