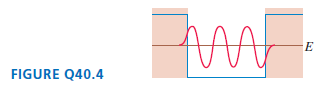
What is the quantum number of the particle in FIGURE Q40.4? How can you tell? E FIGURE
Question:
What is the quantum number of the particle in FIGURE Q40.4? How can you tell?
Transcribed Image Text:
E FIGURE Q40.4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
n 6 be...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
A particle confined to a one-dimensional box has a ground-state energy of 0.4 eV. When irradiated with light of 206.7 nm it makes a transition to an excited state. When decaying from this excited...
-
What is the quantum number of an electron confined in a 3.0-nm-long one-dimensional box if the electrons de Broglie wavelength is 1.0 nm?
-
The contour representation of one of the orbital's for the n = 3 shell of a hydrogen atom is shown below. (a) What is the quantum number l for this orbital? (b) How do we label this orbital? (c) How...
-
A ball is thrown from the roof of a 1 0 0 m high building at a speed of 1 0 m / s making 3 0 angles with the horizontal. A child waiting on the ground at the door of the building falls into a coma...
-
Comment on this statement: "Oh, that beautiful red sunset is just the leftover colors that weren't scattered on their way through the atmosphere."
-
Write an SQL statement to add a CHECK constraint to the PET table so that the weight data recorded in the PetWeight column you added to the table in either question 3.47 or 3.49 is less than 250....
-
Degree of reaction = _____/_____.
-
Scrapper Elevator Company has 20 sales representatives who sell its product throughout the United States and Canada. The number of units sold last month by each representative is listed below. Assume...
-
The total sales of S (in thousands of DVD's) of a certain movie are given by the following formula where t is the number of months since the release of the DVD. Use the formula to answer the...
-
It is March of 2021 and Clayton and Shaleen Miller have come to discuss their tax situation with you. They predict that Clayton will not have to pay taxes for the 2020 year, which they believe will...
-
FIGURE EX40.4 shows the wave function of an electron in a rigid box. The electron energy is 12.0 eV. What is the energy, in eV, of the next higher state? (x) FIGURE EX40.4
-
FIGURE EX40.5 is the probability density for an electron in a rigid box. What is the electron??s energy, in eV? O nm 0.45 nm FIGURE EX40.5
-
What does an individual dot on a scatterplot represent?
-
What type of employee recruitment questions are inappropriate or illegal, and why? Give a couple examples and explain.
-
A Radiation Technologist inherited a substantial sum of money from her grandfather. She wants to use the money to buy into a partnership that owns an imaging group. Is there anything that seems...
-
How The ASTD Competency HR Model, Havard Model and Dave Ulrich's HR Model can apply to employee retention ?
-
What are some of the activities that can take place during each quarter when establishing a Needs Assessment ?
-
Define Group Task Roles. Next, identify four Group Task Roles, and describe the function each role serves in a group. Define Group Maintenance Roles. Next, identify four Group Maintenance Roles, and...
-
At a military base in Texas, Corporal Stan Moneymaker has been offered a wonderful savings plan. These are the salespersons words: During your 48-month tour of duty, you will invest $200 per month...
-
Establish identity. cos( + k) = (-1)k cos , k any integer
-
a. Is there a highest temperature at which ice can exist? If so, what is it? If not, why not? b. Is there a lowest temperature at which water vapor can exist? If so, what is it? If not, why not?
-
What is the diameter of a copper sphere that has the same mass as a 10 cm 10 cm 10 cm cube of aluminum?
-
The cylinder in FIGURE Q18.4 is divided into two compartments by a frictionless piston that can slide back and forth. If the piston is in equilibrium, is the pressure on the left side greater than,...
-
Choose from the following topics to create discussion: Humanity and the Natural Environment Housing and Home Furnishings Privacy and Socializing
-
On December 31, 2020, Pearl Company had $1,101,000 of short-term debt in the for January 21, 2021, the company issued 22,800 shares of its common stock for $42 per brokerage fees and other costs of...
-
During your interview process for the position of program manager for Booker, Tee, and How you are asked to explain the relationship between cost, schedule, and performance? what is your answer to...

Study smarter with the SolutionInn App


