If Experiments 1 and 2 were repeated using a different coloring agent that produces a different color
Question:
If Experiments 1 and 2 were repeated using a different coloring agent that produces a different color when it binds with sulfite, which of the following changes in procedure would be necessary?
F. The new coloring agent should be added to the blank solution, but not to the sample solutions.
G. Both of the coloring agents should be added to the blank solution and to all of the samples.
H. The absorbance of the blank solution made with the new coloring agent should be added to the measured absorbances.
J. The colorimeter should be set to measure at a different wavelength of light.
Experiment 1
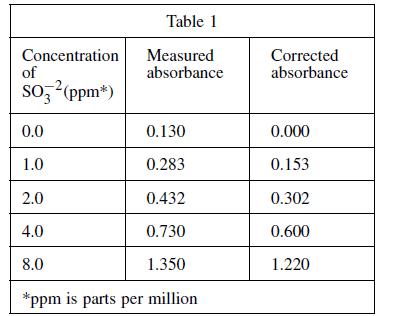
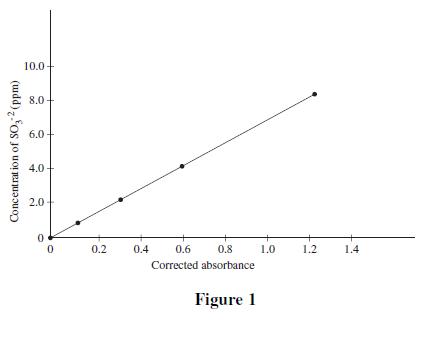
Four solutions, each containing a different amount of sulfite dissolved in H2O were prepared. A coloring agent was added that binds with sulfite to form a red compound that strongly absorbs light of a specific wavelength, and each solution was diluted to 100 mL. A blank solution was prepared in the same manner, but no sulfite was added. A colorimeter (a device that measures how much light of a selected wavelength is absorbed by a sample) was used to measure the absorbance of each solution. The absorbances were corrected by subtracting the absorbance of the blank solution from each reading (see Table 1 and Figure 1).
Experiment 2
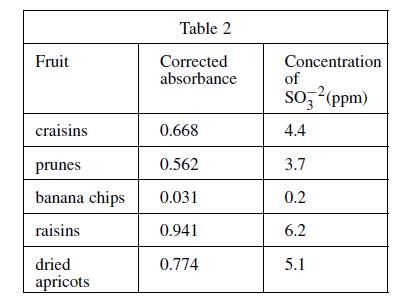
A 100 g fruit sample was ground in a food processor with 50 mL of H2O and the mixture was filtered. The food processor and remaining fruit were then washed with H2O, these washings were filtered, and the liquid was added to the sample solution. The coloring agent was added and the solution was diluted to 100 mL. The procedure was repeated for several fruits, and the absorbances were measured (see Table 2).
Step by Step Answer:






