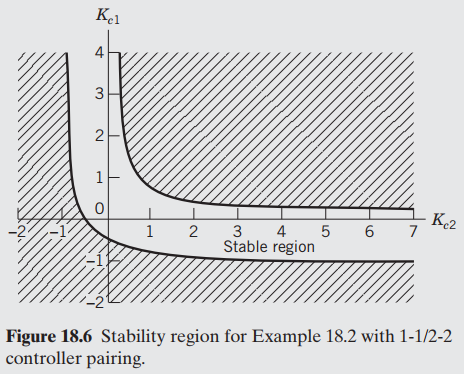
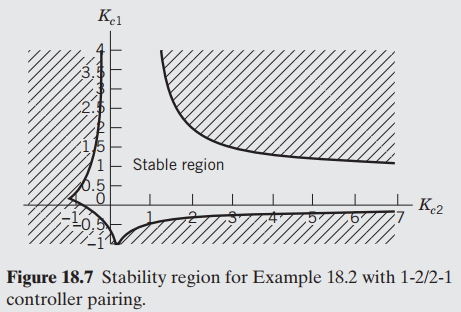
In Figs. 18.6 and 18.7, look at the different pairings of controllers. Which one has the larger
Question:
Transcribed Image Text:
Кл 2 Кг2 4 6 3 5 Stable region Figure 18.6 Stability region for Example 18.2 with 1-1/2-2 controller pairing. 2. 3. Kel (15 Stable region 0.5 K2 Figure 18.7 Stability region for Example 18.2 with 1-2/2-1 controller pairing. AALLO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
1221 controller pairing has a larger stability region compared with 1122 R...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Process Dynamics and Control
ISBN: 978-1119385561
4th edition
Authors: Dale E. Seborg, Thomas F. Edgar, Duncan A. Mellichamp, Francis J. Doyle
Question Posted:
Students also viewed these Physics questions
-
How would the solubility of calcium fluoride be affected by the presence of fluoride ion from another source? What is the solubility of calcium fluoride in a saturated solution of barium fluoride?...
-
With the previous problem in mind, return to the autocorrelation of a sine function, shown in Fig. 11.51. Now suppose we have a signal composed of a great many sinusoidal components. Imagine that you...
-
The specific heat capacity of steel is 450 J/kg C. Show that the amount of heat needed to raise the temperature of a 10-kg piece of steel from 0C to 100C is 450,000 J. How does this compare with the...
-
What is the "ALDI Way" and what was its quest? Take out costs; eliminate complexity Survival; make enough money to pay overhead costs Increase market share; find more prospective customers Build...
-
Johns Hopkins Medicine strives to create a culture in which diversity, inclusion, civility, collegiality, and professionalism are championed through actions, incentives, and accountability. You are a...
-
In December 1995 NatWest, one of the largest British banks, sold its U.S. retail banking operations to Fleet Financial for about $3.5 billion. This price was much less than industry observers had...
-
What are examples of Human Systems Environment threats and their sources?
-
The Furry Friends Foundation is a non-profit organization that finds homes for abandoned animals that are suitable for adoption. FFF began operations with a bequest from a wealthy gentleman who lived...
-
Joe aged 24, and Kate, aged 24, met in college. While studying, Joe worked part-time selling car parts to autobody shops in the GTA. He earned $12,000 a year, for two years. After graduating from...
-
Suppose that the WSClock page replacement algorithm uses a of two ticks, and the system state is the following: where the three flag bits V, R, and M stand for Valid, Referenced, and Modified,...
-
Water (F 1 ) is blended with a stream F 2 with 40% ethanol to make a whiskey product that is 30% ethanol. Assume F 1 = 4 gal/min and F 2 = 4 gal/min. (a) Develop a steady-state material balance model...
-
The thermal effciency of a natural gas boiler versus air/fuel ratio is plotted in Fig. E19.2. Using physical arguments, explain why a maximum occurs. Air-to-fuel stoichiometric ratio Figure E19.2...
-
(a) What is the distinction between cement and concrete? (b) Cite three important limitations that restrict the use of concrete as a structural material. (c) Briefly explain three techniques that are...
-
A 0.00290 kg air-inflated balloon is given an excess negative charge q = -3.00 x 10-8 C by rubbing it with a blanket. It is found that a charged rod can be held above the balloon at a distance of d =...
-
What is the electric force from the rod on the point charge Q? Suppose that Q-17.9 nC and L = 2.81 cm. Break the rod up into four equal pieces and use superposition to find the force. L 2 mC 4L
-
A car of mass 1000 kg is on an icy driveway inclined at an angle of 33. The acceleration of gravity is 9.8 m/s. 0 If the incline is frictionless, what is the acceleration of the car? Answer in units...
-
A linear elastic spring can be compressed 10.0 cm by an applied force of 5.0 N. A 4.5 kg crate of apples, moving at 2.0 m/s, collides with this spring, as shown. What will be the maximum compression...
-
Find the number of atoms in a copper rod with a length of 9.65 cm and a radius of 1.10 cm. The density of copper is 8.96 g/cm. Express the number of atoms to three significant figures.
-
Use a calculator to evaluate expression. Round your answer to three decimal places. cot 28/9
-
Show, if u(x, y) and v(x, y) are harmonic functions, that u + v must be a harmonic function but that uv need not be a harmonic function. Is e"e" a harmonic function?
-
A speed skater moving across friction less ice at 8.0 m/s hits a 5.0-m-wide patch of rough ice. She slows steadily, then continues on at 6.0 m/s. What is her acceleration on the rough ice? Draw a...
-
Sam is recklessly driving 60 mph in a 30 mph speed zone when he suddenly sees the police. He steps on the brakes and slows to 30 mph in three seconds, looking nonchalant as he passes the officer. How...
-
A motorist is traveling at 20 m/s. He is 60 m from a stoplight when he sees it turn yellow. His reaction time, before stepping on the brake, is 0.50 s. What steady deceleration while braking will...
-
Research: What is a sovereign nation, how many sovereign nations are recognized as members by the United Nations and how many sovereign nations are recognized by the US State Department. Compare the...
-
Marigold Corp. purchases a patent for $198,000 on January 2, 2022. Its estimated useful life is 18 years. (a) Prepare the journal entry to record amortization expense for the first year. (Credit...
-
With an interest of $ 2 4 . 3 1 and a principal of $ 3 , 5 0 0 for 1 0 0 days, using the ordinary interest method, the rate is ?

Study smarter with the SolutionInn App


