a. One microliter of 0.1 g/mL 0.1 g/mL benzene is injected into a gas chromatograph using splitless
Question:
a. One microliter of 0.1 μg/mL 0.1 μg/mL benzene is injected into a gas chromatograph using splitless injection. How much benzene is loaded onto the column?
b. How much benzene is loaded onto the column if split injection with a split ratio of 100:1 100:1 is used?
c. If both peak widths are 1 s, 1 s, < what is the mass of carbon entering the detector for splitless and split injection in (a) and (b)?
d. Based on the detection limits in Table 24-3, would a flame ionization detector be sensitive enough to detect the splitless and split injections?
Table 24-3
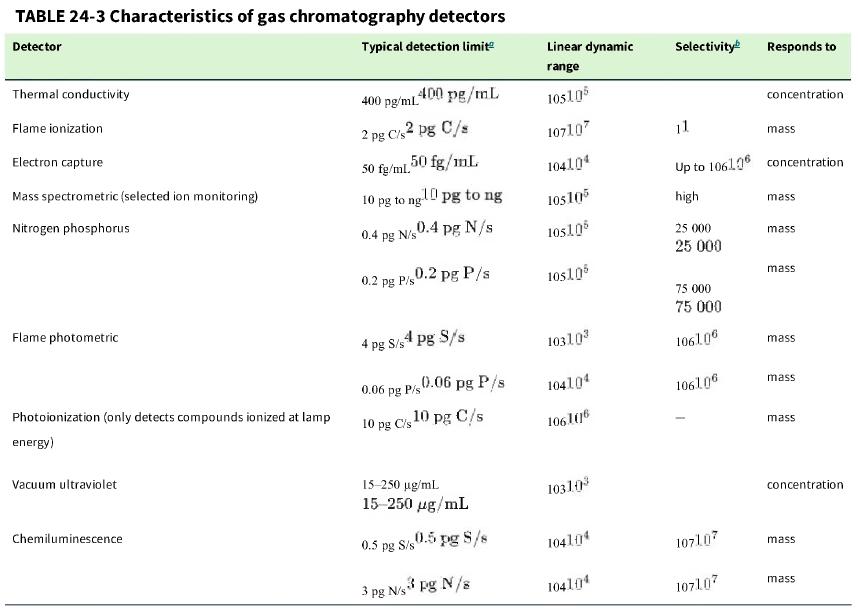
Transcribed Image Text:
TABLE 24-3 Characteristics of gas chromatography detectors Detector Typical detection limit Thermal conductivity Flame ionization Electron capture Mass spectrometric (selected ion monitoring) Nitrogen phosphorus Flame photometric Photoionization (only detects compounds ionized at lamp energy) Vacuum ultraviolet Chemiluminescence 400 pg/mL 2 pg C/s2 pg C/s 50 fg/mL. 10 pg to 0.06 400 pg/mL 50 fg/mL 0.4 pg N/s 0.2 pg P/s 0.2 pg P/s 0.4 pg N/s 4 pg 8/s4 pg S/s pg 3 gl[ pg to ng ng pg 10 pg C/s1 P/s 0.06 pg P/s 10 pg C/s 15-250 µg/mL 15-250 µg/mL 0.5 pg S/s0.5 pg S/s N/s3 pg N/s Linear dynamic range 105105 107107 104104 105105 105105 105105 10310³ 104104 1061016 10310³ 1041014 104104 Selectivity 25 000 25 000 11 Up to 106106 concentration high 75 000 75 000 1061.06 106106 107107 Responds to 107107 concentration mass mass mass mass mass mass mass concentration mass mass
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a The amount of benzene loaded onto the column can be calculated using the equation Amount of ben...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Peak areas and relative detector responses are to be used to determine the concentration of the five species in a sample. The area normalization method described in Problem 32-20 is to be used. The...
-
A chunk of ice of mass ml = 100 g at a temperature tx = 0 C was placed in a calorimeter in which water of mass m1 = 100 g was at a temperature t1. Assuming the heat capacity of the calorimeter to be...
-
A gas chromatograph is an instrument that measures the amounts of various compounds contained in a sample by separating the various constituents. Because different components are flushed through the...
-
1. About 99 percent of the world's population lives outside Canada, but many Canadian companies, especially small businesses, still do not engage in global trade. Why not? Do you think more small...
-
Let the random variable D be defined as in Exercise 7, and let the random variable Q2 be defined by Eq. (11.3.30). a. Show that b. Explain why the random variable Q2/2 will have the 2 distribution...
-
How are standard deviation and variance related? a. The two measures are equal to one another. b. Variance is the square root of the standard deviation. c. Standard deviation is the square root of...
-
Initial cash outflow = \($200,000\) in the current year (year 0), and \($50,000\) in the next year. Cash inflows = \($75,000\) in year 1, \($100,000\) in year 2, \($150,000\) in year 3, and...
-
1. Lastovica Construction is insured under a commercial general liability (CGL) policy. The firm agreed to build a new manufacturing facility for the Smith Corporation. A heavy machine used by...
-
1. How are Michael Porter's forces defined, according to what you have observed in the video? 2. Mention and explain what Michael Porter's 5 forces are. 3. Are there companies in which Michael...
-
1. Give examples of needs, wants, and demands that FedEx customers demonstrate, differentiating these three concepts. 2. Describe FedEx in terms of the value it provides customers. How does FedEx...
-
Efficiency with HPLC Teaching Assistant, an Excel simulation of high performance liquid chromatography. Download the Excel file from SaplingPlus, macmillanlearning.com, or the link in reference...
-
Match descriptions 1-5 1-5 with the band broadening terms in the second list. 1. Depends on radius of open tubular column 2. Not present in an open tubular column 3. Depends on length and radius of...
-
How did government intervention evolve between the first and second halves of the twentieth century?
-
Which of the following are true about Java operators and statements? (Choose three.) A. Both right-hand sides of the ternary expression are evaluated at runtime. B. A switch statement may contain at...
-
Which of the following methods can run without error for at least one SQL query? A. moreChoices() B. stillMoreChoices() C. choices() and stillMoreChoices() D. moreChoices() and stillMoreChoices() E....
-
Given the Electricity annotation, how many lines of the Solar class contain a compiler error? A. One. B. Two. C. Three. D. Four. E. Five. F. Six. G. None of the above. 1: import java.lang.annotation....
-
Which of the following exceptions do not need to be handled or declared by the method in which they are thrown? (Choose three.) A. FileNotFoundException B. ArithmeticException C. IOException D....
-
Which of the following cannot be instantiated directly by the caller using the constructor? (Choose two.) A. Locale B. ResourceBundle C. Locale.Builder D. Properties E. DateTimeFormatter F. HashMap
-
Using the same public organization as in Question 1, discuss the similarities between collective bargaining in this organization and a typical negotiation between a private company and its union?
-
C- Consider the following scenario:- A supermarket needs to develop the following software to encourage regular customers. For this, the customer needs to supply his/her residence address, telephone...
-
A Standard Reference Material is certified to contain 94.6 ppm of an organic contaminant in soil. Your analysis gives values of 98.6, 98.4, 97.2, 94.6, and 96.2 ppm. Do your results differ from the...
-
Nitrite (NO2) was measured by two methods in rainwater and unchlorinated drinking water. The results standard deviation (number of samples) are Sample source Gas chromatography Spectrophotometry Rain...
-
Should the value 216 be rejected from the set of results 192, 216, 202, 195, and 204?
-
In what ways does HR assist managers in handling employee performance issues or conflicts within their teams? What role does HR play in helping managers navigate challenges related to allegations and...
-
Why is A/B testing so useful for marketing, and digital marketing in particular? Give at least two examples of A/B testing for marketing purposes. Give a null and alternative hypothesis for one of...
-
Define the following sets (30 points) (a) X = {(x, y) = R: y = f(x) = 3x + 3y 42} =1 (b) Y={(x, y) = R: y = f(x) = 2x + 2y 42} (c) Z = XUY (d) Prove whether or not each set defined above is convex?

Study smarter with the SolutionInn App


