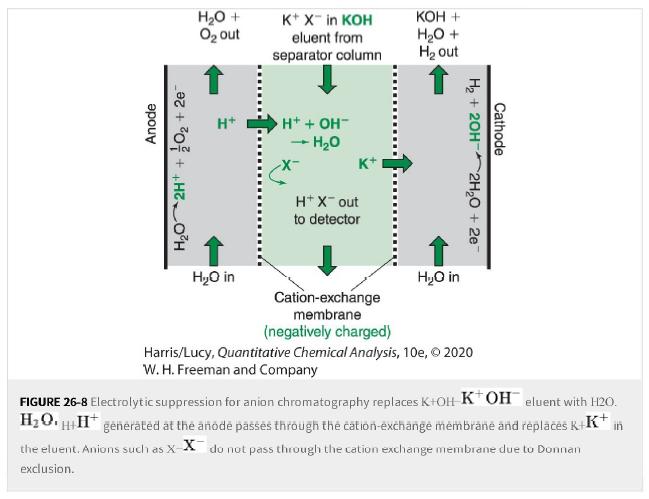
a. The suppressor in Figure 26-8 enables low parts per billion conductivity detection for anions such as
Question:
a. The suppressor in Figure 26-8 enables low parts per billion conductivity detection for anions such as Cl-Cl- and Br-,Br-. but very poor detection limits for anions such as CN-CN- and borate. Explain why.
b. Mixtures of sodium carbonate and bicarbonate can be used as eluent in suppressed-ion anion chromatography. Detection limits are poorer than with hydroxide eluent due to a higher background conductivity. Explain why.
Figure 26-8
Transcribed Image Text:
Anode H₂O 2H+ + O₂ +2e H₂O + O₂ out H+ H₂O in K+ X™ in KOH eluent from separator column H+ + OH™ + H₂O K+ H+ X-out to detector KOH + H₂O + H₂ out H₂O in H₂ + 2OH- Cathode 2H₂O + 2e Cation-exchange membrane (negatively charged) Harris/Lucy, Quantitative Chemical Analysis, 10e, © 2020 W. H. Freeman and Company FIGURE 26-8 Electrolytic suppression for anion chromatography replaces K+OH KOH eluent with H2O. H₂OH+H* generated at the anode passes through the cation-exchange membrane and replaces K+K+ in the eluent. Anions such as X-X do not pass through the cation exchange membrane due to Donnan exclusion.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (11 reviews)
Solution a The suppressor in Figure 268 enables low parts per billion conductiv...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
The carbonate ion (CO32-) can act as either a monodentate or a bidentate ligand. Draw a picture of CO32- coordinating to a metal ion as a bidentate and as a monodentate ligand. The carbonate ion can...
-
Explain why hydroxide ion catalyzes the reaction of piperidine with 2,4-dinitroanisole, but has no effect on the reaction of piperidine with 1-chloro-2,4-dinitrobenzene. piperidine
-
Why are some countries today much poorer than other countries? Are todays poor countries destined to always be poorer than todays rich countries? If so, explain why. If not, explain how todays poor...
-
Please show your work D To be a valid route, it must consist of a sequence of valid move. Each valid move is either going to the right for one block or going down for 1 block. T d) How many valid...
-
For the conditions of Exercise 12 and the data in Table 11.2, carry out a test of the following hypotheses: H0: 1 = 0, H1: 1 = 0.
-
At what interest rate would you be ambivalent about receiving either $50,000 10 years from now or $35,000 today?
-
Explain the key features of project benefits management and how they contribute to enhancing project value.
-
Dillon Awnings manufactures awnings and uses a standard cost system. Dillon allocates overhead based on the number of direct labor hours. The following are the companys cost and standards data:...
-
It has been said that operating managers focus primarily on the income statement, while financial managers focus mainly on the balance sheet. And some say the cash flow statement is the most...
-
How can Clay Potts tactfully explain to Rod Golden that examination of qualitative as well as quantitative documents could be important to delivering an accurate assessment of Fieldss potential to be...
-
Conductivity and contactless conductivity detectors were developed for suppressed capillary ion chromatography. Observed peak heights in millivolts for bromide standards are in the table. a. Use...
-
Low iron concentration (as low as 0.02 nM 0.02 nM) in the open ocean limits phytoplankton growth. Preconcentration is required to determine such low concentrations. Trace Fe3+Fe 3+ from a large...
-
Zero Divisors If a and b are real or complex numbers such that ab = 0. then either a = 0 or b = 0. Does this property hold for matrices? That is, if A and Bare n ( n matrices such that AB = 0. is if...
-
Construct a relative frequency histogram. Use the following information to answer question. Suppose a class of high school seniors had the following distribution of SAT scores in English. SAT score...
-
The light bulbs produced by Edison Lighting Corporation last an average of 300 h. The life of the light bulbs is believed to follow a normal distribution with a standard deviation of 10. A customer...
-
Susan is a one-third partner in Anders Mountain Partners. Last month, she received a non-liquidating cash distribution of $25,000. Her basis in her partnership interest prior to the distribution was...
-
Consider the dilemma you might someday face if you are the chief financial officer of a company that is struggling to maintain a positive cash flow, despite the fact that the company is reporting a...
-
A consumer rights organization wants to find out whether a local dairy farm actually puts 16 oz of milk into the container that is labeled 16 oz. Assume the milk put into the container by the local...
-
Give your assessment of the requirements for Cuba to develop a free-independent trade union. It is essential for a free society?
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
State whether the errors in (a) - (d) are random or systematic: (a) A 25-mL transfer pipet consistently delivers 25.031 0.009 mL. (b) A 10 - mL buret consistently delivers 1.98 0.01 mL when drained...
-
State whether the errors in (a) - (d) are random or systematic: (a) A 25-mL transfer pipet consistently delivers 25.031 0.009 mL. (b) A 10 - mL buret consistently delivers 1.98 0.01 mL when drained...
-
Cheryl, Cynthia, Carmen, and Chastity shot the targets below at Girl Scout camp. Match each target with the proper description. (a) Accurate and precise (b) Accurate but not precise (c) Precise but...
-
In 2019, Rylan Enterprises' net income increased by $2.5 million while its depreciation expense decreased by $500,000, accounts receivable increased by $2,000,000 and accounts payable increased by...
-
Sweeten Company had no jobs in progress at the beginning of March and no beginning inventories. The company has two manufacturing departments-Molding and Fabrication. It started, completed, and sold...
-
The Sky Blue Corporation has the following adjusted trial balance at December 31. Cash Accounts Receivable Prepaid Insurance Notes Receivable (long-term) Equipment Accumulated Depreciation Accounts...

Study smarter with the SolutionInn App


