Ayurveda is a form of medicine practiced in India. In one study, 20%20%. of U.S. and Indian-manufactured
Question:
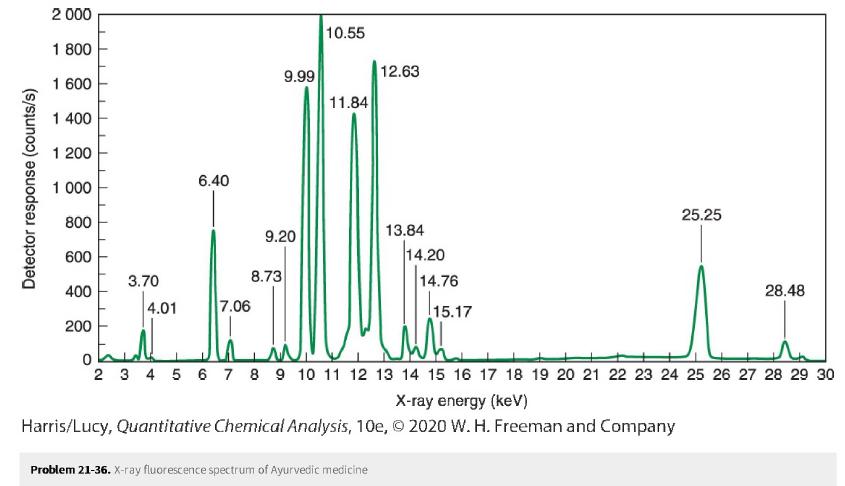
Ayurveda is a form of medicine practiced in India. In one study, 20%20%. of U.S. and Indian-manufactured Ayurvedic medicines purchased through the Internet in 2005 contained detectable levels of several toxic elements. In the spectrum below, identify as many peaks as you can in the X-ray fluorescence spectrum of the Ayurvedic medicine.
Transcribed Image Text:
Detector response (counts/s) 2 000 1 800 1 600 1 400 1 200 1 000 800 600 400 200 3.70 4.01 6.40 7.06 9.99 9.20 8.73 10.55 11.84 12.63 13.84 Problem 21-36. X-ray fluorescence spectrum of Ayurvedic medicine 14.20 14.76 15.17 X-ray energy (keV) Harris/Lucy, Quantitative Chemical Analysis, 10e, 2020 W. H. Freeman and Company 25.25 0 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 28.48
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
Answer The peaks in the Xray fluorescence spectrum are 1 Cadmi...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Holt Industries, Inc. does business in State 1. That state uses federal taxable income as the starting point for calculating state income tax. Interest on state obligations is taxed, but interest on...
-
1. Comment (briefly) on the design of the two costing systems + draw an overview diagram of each system. Explain why you believe / do not believe that the ABC system provides better costing...
-
Ayurveda is a form of medicine practiced in India. In one study, 20% of U.S.- and Indian-manufactured Ayurvedic medicines purchased through the Internet in 2005 contained detectable levels of several...
-
ABC Pty Ltd would like to set up a Virtualisation Platform on their organisation. You have been hired by Company to be their network and system administrator to implement virtualisation for...
-
Show that is as given in Eq. (11.2.5).
-
As of December 31, 2022, Sandy Beach had $9,500,000 in 3 percent serial bonds outstanding. Cash of $509,000 is the debt service fund's only asset as of December 31, 2022, and there are no...
-
Your project is scheduled to last three months and cost $150,000. At the end of the first month, the project is 20% complete. What is the earned value (EV)?
-
Suwon Pharmaceuticals invests heavily in research and development (R&D), although it must currently treat its R&D expenditures as expenses for financial accounting purposes. To encourage investment...
-
Papraika Limited ( also known as Papraika Stores ) is the largest non - food retailer in South Africa and is listed on the Johannesburg Stock Exchange ( JSE ) . The company is the leading retailer of...
-
Each year America.EDU ranks the best paying college degrees in America. The following data show the median starting salary, the mid-career salary, and the percentage increase from starting salary to...
-
Signal in attenuated total reflectance is related to penetration depth. a. Calculate the penetration depth of 1 500 cm -1 1500 cm -1 radiation into an organic sample (n2=1.5)(n 2 =1.5) for a 45450...
-
a. Explain the purpose of the collision cell in Figure 21-28. b. Determination of 28Si 28Si , < experiences isobaric interference from 14N2+ 14N + 2 and background silicon leached from glass...
-
Delta Partnership carries on business in the United States and four other countries. Explain why the ordinary income generated by the foreign business is a separately stated item on Deltas Schedule...
-
How do you feel about your resumeyour present one and the prospect of updating it? When you read it, what makes you feel proud? What makes you feel sad, worried, or inadequate? Try to reframe your...
-
To what extent are you anxious or excited about giving presentations? In what situations are you more comfortable, and in what situations are you more nervous? How do you think your reaction compares...
-
Follow the steps described in the chapter to manage your online reputation. What did you find in a thorough search that included different search engines, name and term combinations, and videos and...
-
After completing the speaking tool (speaking.amynewman.com), share your results with another student in class. Identify five or six strategies you will try before, during, or after a presentation....
-
Join a LinkedIn professional group that interests you. Spend a couple of weeks observing the online discussions, and then post a question and see what, if any, responses you get. Share your posts and...
-
Discuss the advantages and disadvantages of outsourcing or off shoring jobs. Should a worker in today's economy have any reasonable expectation of job security? Explain your reasoning?
-
For each of the following transactions, indicate whether it increases, decreases, or has no effect on the following financial ratios: current ratio, debt-to-equity ratio, profit margin ratio, and...
-
The cumulative formation constant for SnCl2(aq) in 1.0 M NaNO3 is 2 = 12. Find the concentration of SnCl2(aq) for a solution in which the concentrations of Sn2+ and Cl- are both somehow fixed at 0.20...
-
Given the following equilibria, calculate the concentration of each zinc species in a solution saturated with Zn(OH)2(s) and containing [OH-] at a fixed concentration of 3.2 10-7M. Zn(OH)2(s) Ksp =...
-
Although KOH, RbOH, and CsOH have little association between metal and hydroxide in aqueous solution, Li+ and Na+ doform complexes with OH-: Na + + OH- NaoH (aq) K1 = 0.20 Prepare a table like the...
-
1- The binomial distribution N! B(M) = (N-M)!M! describes the probability of placing M identical particles into N equivalent physical sites, with a limit of one particle/site if the probability of a...
-
Margo, a calendar year taxpayer, paid $1,580,000 for new machinery (seven-year recovery property) placed in service on August 1, 2023. Required: a. Assuming that the machinery was the only tangible...
-
CASA (Australia's Civil Aviation Safety Authority) and I CAO SMS (Safety Management Systems) and the alphanumeric risk categorization system for an aviation risk register. *** What I need help with...

Study smarter with the SolutionInn App


