Blank solutions were monitored for vanadium as 51V+ 51 V + using inductively coupled plasma-mass spectrometry with
Question:
Blank solutions were monitored for vanadium as 51V+ 51V+ using inductively coupled plasma-mass spectrometry with the following results.
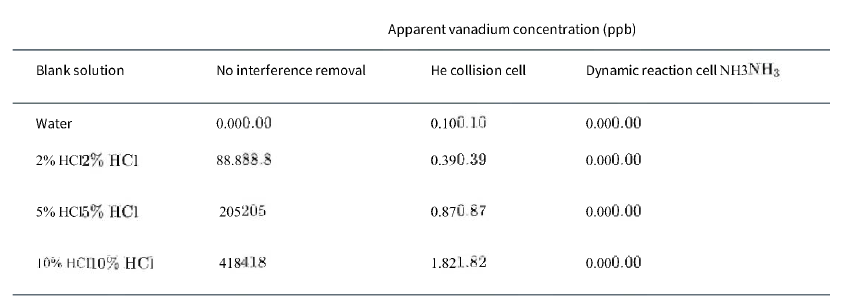
a. Is the HClHCl used to prepare the blank solutions contaminated with vanadium?
b. Would monitoring 50V+50V+ be a better alternative?
c. What acid should be used in place of HClHCl?
Transcribed Image Text:
Blank solution Water 2% HC12% HC1 5% HCI5% HC1 10% HC110% HCI No interference removal 0.000.00 88.888.8 205205 418418 Apparent vanadium concentration (ppb) He collision cell 0.100.10 0.390.39 0.870.87 1.821.82 Dynamic reaction cell NH3NH3 0.000.00 0.000.00 0.000.00 0.000.00
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Answer a No the HClHCl used to prepare the blank s...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Harold conducts a business with the following results for the year: Revenue ......................................................... $20,000 Depreciation on car...
-
Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The...
-
What nonparametric test can be used to compare plasma aldosterone group between Caucasian and African American children? Hypertension Plasma aldosterone has been related to blood-pressure levels in...
-
Starting on January 1, 2024, you put $125 every month into an account with an APR of 4.10%. Your last payment is on December 1, 2032. On January 1 of that year, the balance in the account, rounded to...
-
Moore and McCabe (1999, p. 174) report prices paid for several species of seafood in 1970 and 1980. These values are in Table 11.6. If we were interested in trying to predict 1980 seafood prices from...
-
For the following exercises, find the decomposition of the partial fraction for the nonrepeating linear factors. 5x/x 2 9
-
What is the significance of the 0/100 rule and the 50/50 rule and why would each be used?
-
VIP Limo Service was organized to provide limousine service between the airport and various suburban locations. It has just completed its second year of business. Its trial balance is below. The...
-
Tanner's gross pay for the week is $ 1 9 0 0 . His yearly pay is under the limit for OASDI. Assume that the rate for state and federal unemployment compensation taxes is 6 % and that Tanner's year to...
-
Figure 4-32 shows an EER diagram for a simplified credit card environment. There are two types of card accounts: debit cards and credit cards. Credit card accounts accumulate charges with merchants....
-
Calibration curve. Figure 21-30 describes graphite furnace atomic absorbance determination of silver within individual Daphnia magna. Calibration data at 328.068 nn 328.068 nn is shown below. a....
-
What is the difference between luminescence, chemiluminescence, and bioluminescence?
-
Answer each of the following independent questions. Ignore personal income taxes. 1. Suppose you invest $2,500 in an account bearing interest at the rate of 14 percent per year. What will be the...
-
Provide a brief description of the Apache NetBeans Open Source IDE.
-
In teams of three or four, plan how to divide parts of a presentation. Choose a topic that interests your team: a business idea, a current news item, an upcoming campus event, and so on. Follow these...
-
The Apache NetBeans IDE is composed of two components, which are: . a. NetBeans Platform and NetBeans modules b. NetBeans modules and Java EE c. NetBeans Profiler and GUI design tools d. NetBeans...
-
Watch the videos from Exercise 6 again. This time, observe the delivery skills more closely. What do you find effective and ineffective in each presenters movement, gestures, and eye contact? Stop...
-
A Java Class Library is only a skeleton Java class library without a main class, but it can be executed itself. (True/False)
-
What type of control should an employee have over shares issued under an employee stock ownership plan? For example, should an employee have a right to sell their plan shares at any time or vote...
-
1. What are some current issues facing Saudi Arabia? What is the climate for doing business in Saudi Arabia today? 2. Is it legal for Auger's firm to make a payment of $100,000 to help ensure this...
-
Use Table 6-1 to calculate the pH of pure water at (a) 25C and (b) 100C.
-
An acidic solution containing 0.010 M La3+ is treated with NaOH until La(OH)3 precipitates. At what pH does this occur?
-
Use Le Chtelier's principle and Kw in Table 6-1 to decide whether the autoprotolysis of water is endothermic or exothermic at (a) 25C; (b) 100C; (c) 300C.
-
After you've completed this week's assigned reading, watch the video below. Next, respond to the Discussion questions underneath. Your first post should answer those questions and should be made by...
-
Factorize (x4 - 20x + 100)
-
Paragraph discussing food insecurity in the United States. What agencies in the federal government play an important role in ameliorating this situation? What groups of people are at particular risk?

Study smarter with the SolutionInn App


