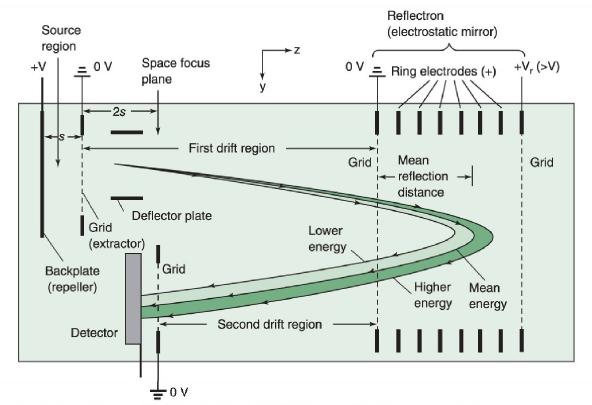
Briefly describe how the time-of-flight mass spectrometer in Figure 22-2 works. What is the benefit of the
Question:
Briefly describe how the time-of-flight mass spectrometer in Figure 22-2 works. What is the benefit of the electrostatic mirror in Figure 22-20?
Figure 22-2

Figure 22-20
Transcribed Image Text:
Grid 1 V₂ = 20 kV to 18 kV Backplate V₂ = 20 kV Sample probe 20 000 V 18 000 V Grid 2 OV Deflector plates Pulsed ultraviolet laser Laser vaporizes samples spot Initial potential Temporary potential after delay time At OVL Backplate Grid 1 20 000 V 18 000 V To high vacuum (10-10 bar) lons with small m/z move faster than ion with large miz -2-m-long field-free drift region Laser pulse Grid 1 potential Af = 0.2-2 μs 10 μs ion Lower ejection grid 1 potential Microchannel plate ion detector -200 V Grid OV Restore grid 1 potential Grid 2 Time Harris/Lucy, Quantitative Chemical Analysis, 10e, © 2020 W. H. Freeman and Company FIGURE 22-2 Schematic diagram of time-of-flight mass spectrometer with matrix-assisted laser desorption/ionization (MALDI) sample introduction and time-delayed ion extraction. -2 kV Gate closed
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
The timeofflight TOF mass spectrometer in Figure 222 works by first ionizing the sample to be analyz...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Briefly describe how the data are used to create profiles of users.
-
Briefly describe how the percentiles are calculated for a data set.
-
Briefly describe how the Bretton Woods system operated.
-
Generate a matrix of random integer temperatures in Fahrenheit from 70 to 100 for 10 weeks (rows) and 7 days per week (columns). The result should look something like this. Assume the first column is...
-
Suppose that both the least-squares line and the least squares parabola were fitted to the same set of points. Explain why the sum of the squares of the deviations of the points from the parabola...
-
The current price of the stock of Mont Tremblant Air is C$100. During each six-month period it will either rise by 11.1 percent or fall by 10 percent (equivalent to an annual standard deviation of...
-
An F-test with 12 degrees of freedom in the numerator and 6 degrees of freedom in the denominator produced a test statistic whose value was 3.42. The null and alternate hypotheses were H0: 1 = 2...
-
You would like to buy an industrial property in Pointe-Claire that has two tenants occupying all of the leasable area. The current owner provides you with the following information. One tenant has...
-
Explain why the managers at Amazon may or may not need to be quality concerned about the same elements of PESTEL
-
The balance sheet items of Kiner Company as of December 31, 2011, follow in random order. You are to prepare a balance sheet for the company, using a similar sequence for assets as illustrated in...
-
The absorbance of a 2.31 x 10 -5 M 2.31x10 -5 M solution of a compound is 0.822 0.822 at a wavelength of 266 nm 266 nm in a 1.000-cm 1.000-cm cell. a. Calculate the molar absorptivity at 266 nm.266...
-
A 100-mL 100-mL sample of hard water containing magnesium and calcium was titrated as illustrated in Figure 18-10. It required 14.59 mL 14.59 mL of 10.83 mM 10.83 mM ethylenediaminetetraacetic acid...
-
Amy Stauffer and her colleagues (2006) examined the interactive effects of victim race and gender on capital murder death sentence outcomes in North Carolina. Their analysis is based on sentences...
-
Find the magnitude of the electric field at a distance of 5 cm away from a line charge on axis with the line charge if the line charge density lambda is 6 uC / m and the length of the line charge is...
-
On January 1, 2024, Sunland issued $4,400,000, 6% bonds. Interest is payable semi-annually on June 30 and December 31. The bonds mature on January 1, 2034. The bonds are sold to yield 12%. Click here...
-
A person whirls a sound tube with a period of 3 rotations per second and a radius of 0.75m. If they hear a pitch of 880 Hz (A on a piano) a. what are the approach and departure speeds of the tube...
-
Criterion Feedback 12/26/22 You have a well thought out overview of internet marketing - good. What are lacking are a few points for why internet marketing improves communication with consumers....
-
Nardin Outfitters has a capacity to produce 20,000 of their special arctic tents per year. The company is currently producing and selling 5,000 tents per year at a selling price of $1,700 per tent....
-
What is the minimum number of times we must toss a fair coin so that the probability that we get at least two heads is at least 0.95?
-
Maria Castigliani is head of the purchasing department of Ambrosiana Merceti, a medium-sized construction company. One morning she walked into the office and said, The main problem in this office is...
-
(a) Following the example of ammonia in Section 7-5, write the equilibria and charge and mass balances needed to find the composition of 0.01 M sodium acetate, which you should abbreviate as Na+A-....
-
Calculate the ionic strength of (a) 0.0087 M KOH and (b) 0.0002 M La(IO3)3 (assuming complete dissociation at this low concentration and no hydrolysis reaction to make LaOH2+).
-
Calculate the ionic strength of (a) 0.0087 M KOH and (b) 0.0002 M La(IO3)3 (assuming complete dissociation at this low concentration and no hydrolysis reaction to make LaOH2+).
-
what ways does cognitive diversity contribute to more effective decision-making processes, and how can organizations leverage this to achieve competitive advantages in a globalized market ?
-
On January 1, 2020 Zappa Enterprises sold 8%, 20-year bonds with a face amount of $1,200,000 for $1,140,000. Interest is payable annually on January 1. Calculate the carrying value of the bond at...
-
2. The polynomial z+az + bz-65 has a factor of (-2-31) Find the values of the real constants and b.

Study smarter with the SolutionInn App


