Calculate E and K for each of the following reactions. (a) I2(s) + 5Br2(aq) + 6H20 =
Question:
Calculate E and K for each of the following reactions.
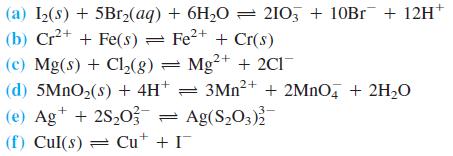
Transcribed Image Text:
(a) I2(s) + 5Br2(aq) + 6H20 = 210, + 10Br + 12H+ (b) Cr2+ + Fe(s) = Fe2+ + Cr(s) (c) Mg(s) + Cl,(g) = Mg²+ + 2C1 (d) 5MNO2(s) + 4H* = 3MN²+ + 2MNO, + 2H2O (e) Ag+ + 2S,03 = Ag(S,O3) (f) Cul(s) = Cu* + I
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
To calculate the standard potential E and equilibrium constant K for each of the given reactions we ...View the full answer

Answered By

Nithesh M
This is Vijaya Chandra, I was graduated from JNTU Engineering College in Mechanical Engineering. I have always shown a keen interest in physics, which is evident from his exceptional academic record in the subject. During my academic years, I was excelled in physics, consistently achieving high grades and performing well in exams. In addition to academic background, I worked as a tutor for 5 years in local village by helping students of all ages and skill levels achieve their goals. As a tutor, I have taught students of all classes for a period of five years, including graduate-level physics and have a deep understanding of the subject and passion for teaching has allowed me to develop unique teaching methods that make complex concepts easy to understand. I am comfortable working with students one-on-one or in small groups, and I am dedicated to helping them build confidence and succeed in their studies. Outside tutoring, I enjoyed playing cricket and surfing internet browser.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Predict the principal organic product of each of the following reactions:
-
Identify the principal organic products of each of the following reactions:
-
Predict the major organic product in each of the following reactions:
-
You are the president of a new, capital-rich firm and you must decide on your firm's targeting approach. Three large subgroups exist in your market that have never had their unique needs served. If...
-
In a taste test of two chocolate chip cookie recipes, 13 out of 18 subjects favored recipe A. Using the sign test, find the significance probability when H1 states that recipe A is preferable.
-
The intangible assets section of Sappelt Company at December 31, 2017, is presented below. Patents ($70,000 cost less $7,000 amortization)......................................$63,000 Franchises...
-
Why are most engineers likely to have experience with deterministic risk and not probabilistic risk?
-
Suppose a ten-year, $1000 bond with an 8% coupon rate and semiannual coupons is trading for a price of $1034.74. a. What is the bonds yield to maturity (expressed as an APR with semiannual...
-
What is the output? int main() { for (int i = 0; i < 3; ++i) { cout < < i; } } cout < < i; return 0;
-
By taking a central flux path, estimate the inductance of the sensor shown in Figure (a) for zero air gap; (b) for a 2 mm air gap. Assume the relative permeability of core and armature is 104 and...
-
In olden days, mercury cells with the following chemistry were used to power heart pacemakers: What is the cell voltage? If the power required to operate the pacemaker is 0.010 0 W, how many...
-
Calculate the voltage of each of the following cells. With the reasoning in Figure 13-8, state the direction of electron flow. (a) Fe(s) | FeBr 2 (0.010 M) || NaBr(0.050 M) | Br 2 (l) | Pt(s) (b)...
-
University of Florida football programs are printed 1 week prior to each home game. Attendance averages 90,000 screaming and loyal Gators fans, of whom two-thirds usually buy the program, following a...
-
Revit comes with many predefined doors and windows. A) True B) False
-
Fill in the blank field in this text: The icon with the floppy disk picture allows you to [1]__________ a project file.
-
Skylights need to be rotated to align with the plane, or pitch, of the roof. A) True B) False
-
When using the Mirror tool, you occasionally need to draw a temporary line that represents the Axis of Reflection. A) True B) False
-
Fill in the blank field in this text: Use the [1]__________tool to adjust the ceiling grid location if a ceiling tile is less than half its normal size.
-
The owner of newly formed Lake of the Woods Air Taxi, a friend of your family, knows you are taking an accounting course and asks for some advice. Mr. Linden tells you that he is pretty good at...
-
What tools are available to help shoppers compare prices, features, and values and check other shoppers opinions?
-
Nitric oxide (NO) is a cell-signaling agent in physiologic processes including vasodilation, inhibition of clotting, and inflammation. A sensitive chromatography-mass spectrometry method was...
-
Equation 22-33 contains terms (A, B, and C) describing three band-broadening mechanisms. (a) Which term is 0 for an open tubular column? Why? (b) Express the value of B in terms of measurable...
-
As the inside radius of an open tubular column is decreased, the maximum possible column efficiency increases and sample capacity decreases. For a thin stationary phase that equilibrates rapidly with...
-
How would Stephanies investing decisions be affected if she were 35 years old? If she were 50 years old? Amount of Insurance Needed. Considering the information in the previous problem, how much life...
-
Jamie Lee and Ross have been hearing many stories recently about acquaintances who are passing away without leaving a will, which made Jamie Lee and Ross anxious to review their estate plan with an...
-
What are the companys top risks, how severe is their impact and how likely are they to occur?

Study smarter with the SolutionInn App


