Consider the following equilibria in aqueous solution: (a) Calculate the numerical value of the equilibrium constant for
Question:
Consider the following equilibria in aqueous solution:
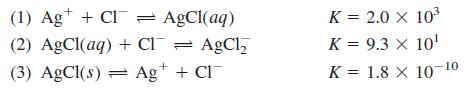
(a) Calculate the numerical value of the equilibrium constant for the reaction
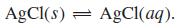
(b) Calculate the concentration of AgCl(aq) in equilibrium with excess undissolved solid AgCl.
c) Find the numerical value of K for the reaction
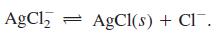
Transcribed Image Text:
(1) Ag* + Cl = AgCl(aq) (2) AgCl(aq) + Cl¯ = AgCl, K = 2.0 X 103 %3D K = 9.3 x 10' (3) AgCl(s) = Ag+ + Cl K = 1.8 X 10-10
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a The overall reaction is Ag Cl Ag Cl We can write the equilibrium expression for this reaction as K ...View the full answer

Answered By

FELIX NYAMBWOGI
I have been tutoring for over 5 years, both in person and online. I have experience tutoring a wide range of subjects, including math, science, English, and history. I have also worked with students of all ages, from elementary school to high school.
In addition, I have received training in effective tutoring strategies and techniques, such as active listening, questioning, and feedback. I am also proficient in using online tutoring platforms, such as Zoom and Google Classroom, to effectively deliver virtual lessons.
Overall, my hands-on experience and proficiency as a tutor has allowed me to effectively support and guide students in achieving their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Consider the equilibrium Calculate the equilibrium constant Kp for this reaction, given the following information (at 298 K):
-
Consider the system A(g) B(g) at 25oC. a. Assuming that GoA = 8996 J/mol and GoB = 11,718 J/ mol, calculate the value of the equilibrium constant for this reaction. b. Calculate the equilibrium...
-
Consider the following reaction run at standard conditions: Al(s) + Fe2+(aq) Fe(s) +Al3+(aq) a. Calculate the standard cell potential for this cell from standard free energies of formation (see...
-
Assume that you are purchasing shares in a company in the variety store and gas bar supply business. Suppose you have narrowed the choice to BFI Trading Ltd. and Lin Corp. and have assembled the...
-
Sue is 30 years old and is president and a 51% shareholder of C Corporation. She informs you that C Corporation has 10 shareholders, all unrelated. Other than herself, no shareholder owns more than...
-
Hartman Motors has $18 million in assets, which were financed with $6 million of debt and $12 million in equity. Hartmans beta is currently 1.3, and its tax rate is 35%. Use the Hamada equation to...
-
Some might say that coding is not as important as analysis or design since programming errors can be identified and corrected easily. Do you agree with this statement? Why?
-
A study dealing with health care issues plans to take a sample survey of 1500 Americans to estimate the proportion who have health insurance and the mean dollar amount that Americans spent on health...
-
Units Beginning work in process inventory 32,000 Percent Complete 65% Percent Complete 35% Units started this period 410,000 Completed and transferred out 412,000 Ending work in process inventory...
-
Jon Williams, CPA, is in the middle of the real- life soap opera, Taxing Days of Our Lives. The Cast of Characters Oneway Corporation is Williamss audit and tax client. The three directors are the...
-
The U.S. Department of Agriculture provided homogenized baby food samples to three labs for analysis.3 Results agreed well for protein, fat, zinc, riboflavin, and palmitic acid. Results for iron were...
-
Reaction 6-7 is allowed to come to equilibrium in a solution initially containing 0.0100 M BrO - 3 , 0.0100 M Cr 3+ , and 1.00 M H + . To find the concentrations at equilibrium, we can construct a...
-
Mr. Carl Lipke is the marketing VP for a propane gas distributor. He would like to have a forecast of sales on a quarterly basis, and he has asked you to prepare a time-series decomposition model....
-
Suppose Fred has an income of $5,000 per year, and Harry has an income of $105,000 per year. If we tax $50,000 from Harry to give to Fred, will this represent a Pareto improvement for society? Why or...
-
If physician fees are fi xed so that they do not adjust to changes in supply, explain how a fi rm whose demand curve slopes downward would react if the fi xed fee were lowered, perhaps by the...
-
What is the Medicare PPS program under DRGs? How has the Medicare PPS payment under DRGs affected hospital practices? Length of stay? Quality of care? Financial condition?
-
Consider Figure 14.1. Suppose that a hospital has the ability to be a perfect price discriminator in the private market, that is, it can charge every private patient the maximum that the patient is...
-
Suppose that insurers monitored all health care payments to determine whether the services were appropriate. Would you expect to see more or less tendency toward SID?
-
An airline analyst wishes to estimate the proportion of all American adults who are afraid to fly because of potential terrorist attacks. To estimate this percentage, the analyst decides to survey...
-
-4 1 9. Let A = Find A-1, (A") and verify that (A")= (A-1)".
-
Fractional composition in a tetraprotic system. Prepare a fractional composition diagram analogous to Figure 9-4 for the tetraprotic system derived from hydrolysis of Cr3+: Cr3+ + (H2O) Cr(OH)2+ +...
-
What is the difference between the isoelectric pH and the isoionic pH of a protein with many different acidic and basic substituents?
-
Consider the diprotic acid H2A with K1 = 1.00 10-4 and K2 = 1.00 10-8. Find the pH and concentrations of H2A, HA-, and A2- in (a) 0.100 M H2A; (b) 0.100 M NaHA; (c) 0.100 M Na2A.
-
Discuss the dynamic organization of the cytoskeleton and its pivotal role in cellular motility, intracellular transport, and structural integrity .
-
Would you support the idea of a government issued Digital currency? Why ? and why not?
-
To protect her savings against further inflation and to help her prepare for a healthy financial future, Hanna Lind deposits $9,100 in an investment account earning 6% interest compounded quarterly....

Study smarter with the SolutionInn App


