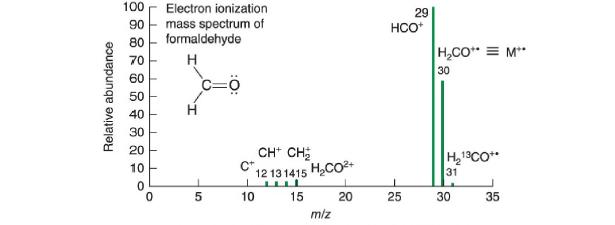
Formaldehyde has a nominal (integer) mass of 30 Da, 30 Da, but its mass spectrum in Figure
Question:
Formaldehyde has a nominal (integer) mass of 30 Da, 30 Da, but its mass spectrum in Figure 22-7 has seven peaks at different values of m/z m/z. Explain the origin of each peak. Draw a Lewis dot structure of each ion to decide which ones have an unpaired electron.
Figure 22-7
Transcribed Image Text:
Relative abundance 100 Electron ionization mass spectrum of formaldehyde H 90 80 70 60 50 40 30 20 10 0 0 H 5 =0 CH CH₂ 12131415 HCO 15 C+ I 10 m/z 20 29 HCO+ 25 H₂CO = M** 30 H₂13CO** 31 30 35
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
Answer The seven peaks in the mass spectrum of formaldehyde correspond to different ionic ...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Draw a Lewis dot structure for each of the following compounds: a. CH 3 CH 2 OH b. CH 3 CN
-
Draw the Lewis electron dot diagram for each ion. a. In+ b. Br
-
Draw the Lewis electron dot diagram for each ion. a. Fe2+ b. N3
-
2. Using the data below, create the project schedule using normal times. Determine the order in which you would crash the project one day, two days, and so on until it is in an all-crash mode....
-
Consider again the conditions of Exercise 7, and let = 30 + c11, where c1 is a constant. Determine an unbiased estimator For what value of c1 will the M.S.E. of be smallest?
-
Hors dAge Cheeseworks has been paying a regular cash dividend of $4 per share each year for over a decade. The company is paying out all its earnings as dividends and is not expected to grow. There...
-
Little People Learning Center (LPLC) has assembled the following data for the year ended June 30, 2005. Prepare the operating activities section of LPLC's statement of cash flows for the year ended...
-
Bolster Foods' (BF) balance sheet shows a total of $25 million long-term debt with a coupon rate of 8.50%. The yield to maturity on this debt is 8.00%, and the debt has a total current market value...
-
Hinsdale Corp. issues 1,000 shares of $3 par value common stock at $20 per share. Later in the year, the company repurchased 200 shares at a cost of $22 per share. 1. Record the journal entry for the...
-
Determine the nodal displacements and the element stresses, including principal stresses, for the thin plate of Section 6.5 with a uniform shear load (instead of a tensile load) acting on the right...
-
Explain why formation of volatile arsine (AsH 3 ) (AsH 3 ) improves detection limits versus direct nebulization of an arsenic solution.
-
Literature problem. Zinc is an essential micronutrient. It is estimated that 22 billon people are zinc deficient. B. J. Stevens et al. (Latin for and others) wrote a Technical Note (J. Anal. Atom....
-
Let {u, v, w} be a linearly independent set of vectors in a vector space V. (a) Is {u + v, v + w, u + w} linearly independent? Either prove that it is or give a counterexample to show that it is not....
-
1. How should Polaris balance the pursuit of new technologies with scaling capabilities to work with existing technologies? 2. How should the company balance its client portfolio? 3. What would be...
-
Consider the active low pass filter of the following figure. Use Matlab to find and plot the magnitude response, given that: 4 2 kilohms R For Z = 50 , = 1 K, R, = 10 , R, = 1K, R = 2 K and C = 0.1...
-
As a result of malpractice suits and legal actions, it is generally recognized that a professional relationship or even the perception that the relationship is a professional one constitutes the...
-
The concept of thermionic emission is the basis of modern X-ray tubes, which are the workhorse of a medical X-ray image. In this process, a filament (a piece of metal in a vacuum) is heated by the...
-
1. Given its product, resources, and/or general corporate culture, how would the company define effectiveness? How would the company define efficiency? 2. What is the organization's vision statement?...
-
a) Derive a formula for ni=1 t2l, where t2i denotes the 2ith triangular number for 1 < i < n. b) Determine 100i=1 t21. c) Write a computer program to check the result in part (b).
-
A police officer pulls you over and asks to search your vehicle because he suspects you have illegal drugs inside your car. Since he doesn't have reasonable suspicion to search your car, legally he...
-
To evaluate the equilibrium constant in Equation 6-2, we must express concentrations of solutes in mol/L, gases in bars, and omit solids, liquids, and solvents. Explain why.
-
BaCl2 H2O(s) loses water when it is heated in an oven: BaCI2 H2O (s) BaCl2(s) + H2O(g) H = 63.11 kJ/mol at 25C S = + 148J(K mol) at 25C (a) Write the equilibrium constant for this reaction....
-
BaCl2 H2O(s) loses water when it is heated in an oven: BaCI2 H2O (s) BaCl2(s) + H2O(g) H = 63.11 kJ/mol at 25C S = + 148J(K mol) at 25C (a) Write the equilibrium constant for this reaction....
-
For 2012, Tri-Comic Company initiated a sales promotion campaign that included the expenditure of an additional $50,000 for advertising. At the end of the year, Lumi Neer, the president, is presented...
-
Initially at rest, an egg is released from the top of a tall building. After a brief delay, a second egg is hurled down from the same height at a rate of 6.15 m/s. If both eggs splat on the ground at...
-
(a) Can a single isolated stationary object of mass 4 spontaneously explode into two objects each of rest mass greater than 2? Explain your reasoning. (b) Can a single isolated stationary object of...

Study smarter with the SolutionInn App


