Succinic acid dissociates in two steps: K1 H,H,C ,, + H* %3| || OCCH,CH,CO + H* ,
Question:
Succinic acid dissociates in two steps:
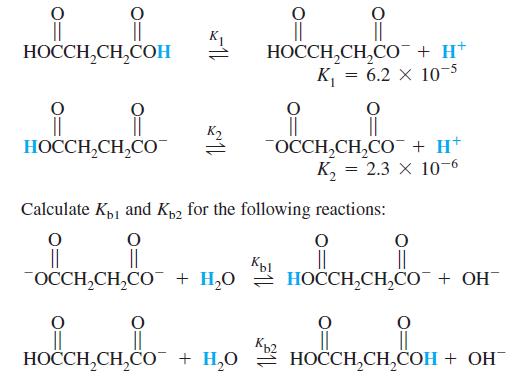
Transcribed Image Text:
K1 НОССH,СH,CОН НОССН,СН,СО + H* %3| || OCCH,CH,CO¯ + H* К, 3 2.3 х 10-6 HOCCH,CH,CO Calculate Kp1 and Kp2 for the following reactions: || OCCH,CH,CO¯ + H,0 = HOCCH,CH,CO + OH Кы Kp2 HOCCH,CH,CO¯ + H,0 2 HOCCH,CH,COH + OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
K1 1 HOOC CHzz COOH HOOC CHzz COOH K 62 x 105 On reversing the reaction K1 HOOC CH22 COOH HOOC C...View the full answer

Answered By

Punit Tiwari
I'm a regular faculty in a well established IIT JEE preparation institute of kota. I'm teaching chemistry from last 4 years for board, medical and engineer entrance exams. Chemistry for me is the dream world I experience every moment. Being an engineer helps me to regenerate the subject in a unique way.
My teaching style associates with the learn with fun technique. I help the students to visualize the chemistry in real world. Come and experience with me the beautiful aura of chemistry and beat the rest of world with your imagination
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Give the structure of the ester formed when (a) Succinic acid reacts with a large excess of diazomethane in ether. (b) Benzoic acid reacts with allyl bromide and K2CO3 in acetone.
-
How would you design a regulatory system to make Escherichia coli use succinic acid in preference to glucose? How could you modify it so that E. coli prefers to use succinic acid in the light but...
-
Show how you might synthesize β-aminopropionic acid from succinic anhydride. (β-Aminopropionic acid is used in the synthesis of pantothenic acid, a precursor of coenzyme A.)...
-
Torres Investments acquired $160,000 of Murphy Corp., 5% bonds at their face amount on October 1, Year 1. The bonds pay interest on October 1 and April 1. On April 1, Year 2, Torres sold $60,000 of...
-
What are the central components of a revenue ruling?
-
You have a line of credit loan with Scotiabank. The initial loan balance was $6000.00. Payments at $2000.00 and $1500.00 were made after 5 months and 10 months, respecively. At the end of one year,...
-
What is the relationship of technology to the information system architecture?
-
On January 1, 2011, Rapid River Realty sold a tract of land to three doctors as an investment. The land, purchased 10 years ago, was carried on Rapid Rivers books at a value of $210,000. Rapid River...
-
Helping Hand Hobby Stores has just paid an annual dividend of $1.20 a share. The company's policy is to increase the dividend at an annual rate of 5.4 percent. What's the current value of the stock...
-
In presenting a report to a group of grocery store managers, a researcher stated the following: "The data from the judgment sample of ten grocery stores were analyzed, and the results show that the...
-
If each compound is dissolved in water, will the solution be acidic, basic, or neutral? (a) Na Br (e) (CH3),N CI (b) Na CH,CO, (f) (CH,),N*O-co, (c) NH CI (g) Fe(NO3)3 (d) K3PO4
-
Histidine is a triprotic amino acid: What is the value of the equilibrium constant for the reaction CO,H -NH CO, -NH HC-CH, K, = 3 x 102 HC-CH, NH, NH, , 3 8.5 10-7 CO, -NH CO NH HC-CH2 , 3 4.6 10...
-
Repeat the preceding exercise using the Wilcoxon signed-ranks test. Preceding Exercise Use a 0.05 significance level with the indicated test. If no particular test is specified, use the appropriate...
-
Explain why the indifference curves in Figure 15.2 are positively sloped. Net income A B 0 U1 mQo+ml E E2 U2 m'Q+m'I I IE,TE2 Inducement Figure 15.2 Physician's Response to Reduced Rate of Profit
-
Of the Newhouse and the physicians cooperative models, which nonprofi t hospital is likely to produce more quantity and quality in equilibrium with barriers to entry? In longrun equilibrium, with...
-
Give examples of ways in which labor and capital can be substituted for each other in the production of health services.
-
In 2004, Congressman Dennis Kucinich proposed the Free Market Drug Act. This legislation would have removed patent protection on drugs developed with public funds and given control over...
-
According to clinical research, nearly one-half of the care provided in the United States falls short of recommended treatment protocols. Discuss how imperfect and asymmetric information contribute...
-
A fast food franchise tracked the number of errors that occurred in customers' orders. These included wrong menu item, wrong size drink, lack of condiments, wrong price total, and so on. Some orders...
-
On the basis of the details of the following fixed asset account, indicate the items to be reported on the statement of cashflows: ACCOUNT Land ACCOUNT NO. Balance Date Item Debit Credit Debit Credit...
-
A 100.0-mL aliquot of 0.100 M diprotic acid H2A (pK1 4.00, pK2 8.00) was titrated with 1.00 M NaOH. Find the pH at the following volumes of base added and make a graph of pH versus Vb: Vb 0, 1, 5, 9,...
-
Calculate the pH at 10.0-mL intervals (from 0 to 100 mL) in the titration of 40.0 mL of 0.100 M piperazine with 0.100 M HCl. Make a graph of pH versus Va.
-
Consider the titration of 50.0 mL of 0.100 M sodium glycinate, H2NCH2CO2Na, with 0.100 M HCl. (a) Calculate the pH at the second equivalence point. (b) Show that our approximate method of...
-
4. The water in Earth's atmosphere blocks most of the infrared waves coming from space. In order to observe light of this wavelength, the Kuiper Airborne Observatory has been developed. The...
-
What amount of gain should Pharoah recognize on the exchange assuming lack of commercial substance?
-
If R1=7 Q, R2=3 Q, R3=9 Q, V= 12 V, and V2=8 V. Find the current (in A) that pass through R. R R R V V 2 +

Study smarter with the SolutionInn App


