The spreadsheet lists molar absorptivities of three dyes and the absorbance of a mixture of the dyes
Question:
The spreadsheet lists molar absorptivities of three dyes and the absorbance of a mixture of the dyes at visible wavelengths.
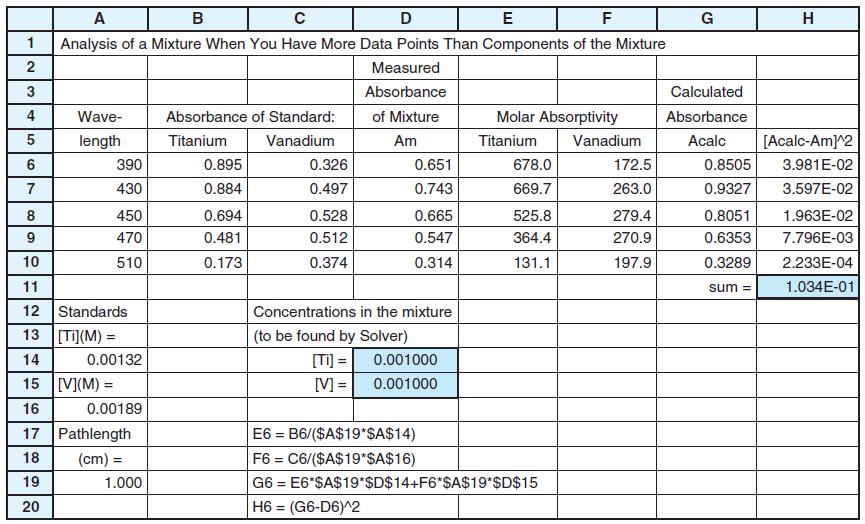
Use the least-squares procedure in Figure 18-3 to find the concentration of each dye in the mixture.
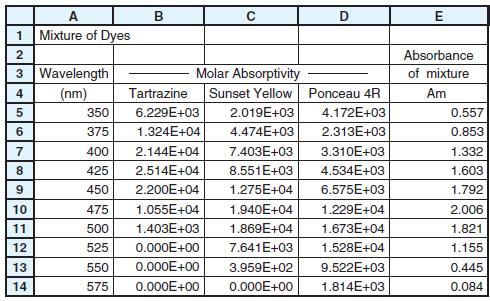
Figure 18-3
Transcribed Image Text:
A Mixture of Dyes B 1 Absorbance 3 Wavelength (nm) Molar Absorptivity Sunset Yellow Ponceau 4R of mixture 4 Tartrazine Am 350 6.229E+03 2.019E+03 4.172E+03 0.557 6 375 1.324E+04 4.474E+03 2.313E+03 0.853 7 400 2.144E+04 7.403E+03 3.310E+03 1.332 8. 425 2.514E+04 8.551E+03 4.534E+03 1.603 9. 450 2.200E+04 1.275E+04 6.575E+03 1.792 10 475 1.055E+04 1.940E+04 1.229E+04 2.006 11 500 1.403E+03 1.869E+04 1.673E+04 1.821 12 525 0.000E+00 7.641E+03 1.528E+04 1.155 13 550 0.000E+00 3.959E+02 9.522E+03 0.445 14 575 0.000E+00 0.000E+00 1.814E+03 0.084
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The spreadsheet gives the product b for four pure compounds and a mixture at infrared wavelengths. Modify Figure 18-5 to solve four equations and find the concentration of each compound. You can...
-
The molar absorptivity for the complex formed between bismuth (III) and thiourea is 9.32 ( 103 L cm21 mol-1 at 470 nm. Calculate the range of permissible concentrations for the complex if the...
-
The logarithm of the molar absorptivity for acetone in ethanol is 2.75 at 366 nm. Calculate the range of acetone concentrations that can be used if the absorbance is to be greater than 0.100 and less...
-
Which one of the following statements related to investigations of workplace harassment complaints under Ontario's OHSA is true? a) As a rule, such investigations should be completed with 120 days of...
-
We all typically go to the shortest line in the grocery store. Data were collected on the number of carts ahead in line and the total time to check out (minutes), including time in line, on five...
-
Pretzelmania, Inc., issues 7%, 15-year bonds with a face amount of $70,000 for $64,008 on January 1, 2021. The market interest rate for bonds of similar risk and maturity is 8%. Interest is paid...
-
Meddevco (name changed) is a large multinational corporation that operates in the medical devices sector. The firm employs around 33,000 people in five divisions and has operations in 120 countries....
-
The equivalent of a new kindergarten class is born every day at Orlandos Arnold Palmer Hospital. With more than 12,300 births in 2005 in a hospital that was designed in 1989 for a capacity of 6,500...
-
A firm has 10,000,000 shares of common stock outstanding, each with a market price of $15.00 per share. It has 80,000 bonds outstanding, each selling for $1020. The bonds mature in 15 years, have a...
-
Kevin and June Park (K and J) are in the process of buying a new house. Three houses, A, B, and C, are available. The Parks have agreed on two criteria for the selection of the houseamount of yard...
-
This problem can be worked with Equations 18-6 on a calculator or with the spreadsheet in Figure 18-5. Transferrin is the iron-transport protein found in blood. It has a molecular mass of 81 000 and...
-
Compound P, which absorbs light at 305 nm, was titrated with X, which does not absorb at this wavelength. The product, PX, also absorbs at 305 nm. Absorbance was measured in a 1.000-cm cell, and the...
-
Hybrid recommendation engines utilize two or more filtering strategies to create recommendations. Describe the four different approaches to creating a hybrid system.
-
TCP is a very symmetric protocol, but the client/server model is not. Consider an asymmetric TCP-like protocol in which only the server side is assigned a port number visible to the application...
-
An organization has been assigned the prefix 200.1.1/24 (a class C) and wants to form subnets for four departments, with hosts as follows: There are 145 hosts in all. (a) Give a possible arrangement...
-
Learn about a key escrow, or key surrender, scheme (for example, Clipper). What are the pros and cons of key escrow?
-
In stop-and-wait transmission, suppose that both sender and receiver retransmit their last frame immediately on receipt of a duplicate ACK or data frame; such a strategy is superficially reasonable...
-
You are an Internet service provider; your client hosts connect directly to your routers. You know some hosts are using experimental TCPs and suspect some may be using a greedy TCP with no congestion...
-
University Cycle Shop had trouble collecting its account receivable from Matt Reid. On January 19, University finally wrote off Reid's $2,400 account receivable. University turned the account over to...
-
The Taylor's series expansion for cosx about x = 0 is given by: where x is in radians. Write a user-defined function that determines cosx using Taylor's series expansion. For function name and...
-
Neutral lysine can be written HL. The other forms of lysine are H 3 L 2+ , H 2 L + , and L + . The isoelectric point is the pH at which the average charge of lysine is 0. Therefore, at the...
-
We will abbreviate malonic acid, CH 2 (CO 2 H) 2 , as H2M. Find the pH and concentrations of H 2 M, HM + , and M 2+ in (a) 0.100 M H 2 M; (b) 0.100 M NaHM; (c) 0.100 M Na 2 M.
-
Starting with the fully protonated species, write the stepwise acid dissociation reactions of the amino acids glutamic acid and tyrosine. Be sure to remove the protons in the correct order. Which...
-
Find all the complex numbers z such that z += 0. Is the set open or closed? {zC: z*+i=0}
-
For each of the following matrices A Maxn (R), test A for diagonal- izability, and if A is diagonalizable, find an invertible matrix Q and a diagonal matrix D such that Q-1AQ = D. (a) (63) 2 1 3 (b)...
-
A summary of two stocks is shown. 52W high 52W low Name of Stock Symbol High Low Close 37.18 29.39 Zycodec ZYO 11.76 7.89 Unix Co UNX 39.06 32.73 34.95 16.12 12.11 15.78 Last year, a stockholder...

Study smarter with the SolutionInn App


