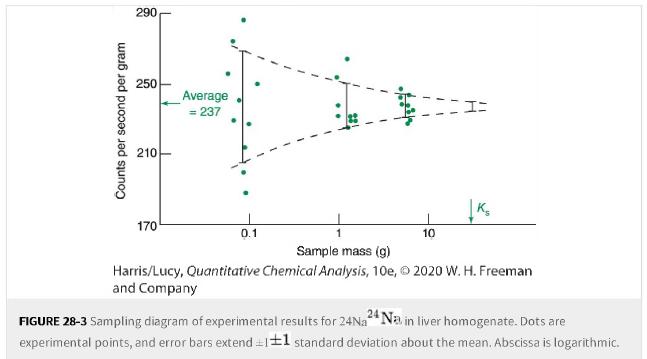
What mass of sample in Figure 28-3 is expected to give a sampling standard deviation of 6%?6%?
Question:
What mass of sample in Figure 28-3 is expected to give a sampling standard deviation of ±6%?±6%?
Figure 28-3
Transcribed Image Text:
Counts per second per gram 290 250 210 170 Average = 237 0.1 10 K₂ Sample mass (g) Harris/Lucy, Quantitative Chemical Analysis, 10e, 2020 W. H. Freeman and Company FIGURE 28-3 Sampling diagram of experimental results for 24Na 24 Na in liver homogenate. Dots are experimental points, and error bars extend = 1±1 standard deviation about the mean. Abscissa is logarithmic.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
Calculations The mass of sample in Figure 283 is expected to give ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Louvre Inc. bought a business that is expected to give a 25% annual rate of return on the investment. Of the total amount paid for the business, $75,000 was deemed to be goodwill, and the rest was...
-
Which of the following would be expected to give a positive test with Benedict's reagent? Why? (a) L-Arabinose (b) 1,3-Dihydroxyacetone (c) D-Fructose (d) Lactose (e) Amylose
-
What are the mean and standard deviation of the sampling distribution of x ? What are the mean and standard deviation of the sampling distribution of p?
-
1. A projectile is launched in a vertical plane, at an angle 0 with initial velocity vo. It must be caught in a frictionless circular tube of radius R in such a way that the trajectory of the...
-
Suppose that X1, . . . , Xm form a random sample from a continuous Distribution for which the p.d.f. f (x) is unknown; Y1, . . . , Yn form an independent random sample from another continuous...
-
The farmer does not avoid risk by selling wheat futures. If wheat prices stay about $2.80 a bushel, then he will actually have lost by selling wheat futures at $2.50. Is this a fair comment?
-
In an experiment to determine the effect of ambient temperature on the emissions of oxides of nitrogen (NOx) of diesel trucks, 10 trucks were run at temperatures of 40F and 80F. The emissions, in...
-
September Company has five salaried employees. Your task is to use the following information to prepare a payroll register to calculate net pay for each employee: Assume the following:1. FICA OASDI...
-
Question 3 (10 marks) Dishwasher Company currently earns an annual contribution of N$2 880 000 from the sale of 90 000 bottles of dishwashing liquid. Fixed costs are N$800 000 per annum. The...
-
Carrie DLake, Reed A. Green, and Doug A. Divot share a passion for golf and decide to go into the golf club manufacturing business together. On January 2, 2018, DLake, Green, and Divot form the...
-
Cloud-point extraction. Traces of lead in drinking water cause adverse health effects. The micelle-forming surfactant Triton X-114 (plus the co-surfactant cetyl trimethylammonium bromide) and a crown...
-
In the chapter opener, trace metals in teeth and bone provide information about Otzi the Icemans diet and environment as a child and adult. The inorganic mineral matrix of teeth and bone is...
-
A: Suppose a firm employs labor and capital k to produce output x using a homothetic, decreasing returns to scale technology. (a) Suppose that, at the current wage w, rental rate r and output price...
-
In this week's reading, I was able to learn that there are lots of different types of advanced directives but the most popular ones are directives to physicians and family (living will), medical...
-
Back in 2003, Nicholas Carr set off a firestorm of controversy in the tech world with his Harvard Business Review article, "IT Doesn't Matter" (OK, maybe "firestorm" is an overstatement, but it was a...
-
Please respond to this discussion post below detailed and throughly, Reducing minimum requirements for law enforcement recruitment can be a double-edged sword. On one side, it could open the door to...
-
Prior to any questioning, the person must be warned that he [or she] has the right to remain silent, that any statement he [or she] does make may be used as evidence against him [or her], and that he...
-
Over a time interval of 1.80 years, the velocity of a planet orbiting a distant star reverses direction, changing from +20.0 km/s to -20.1 km/s. Find (a) the total change in the planet's velocity (in...
-
Consider the membership table (Table 3.7). If we are given the condition that A B, then we need consider only those rows of the table for which this is true - rows 1, 2, and 4, as indicated by the...
-
In July 2013, cnet.com listed the battery life (in hours) and luminous intensity (i. e., screen brightness, in cd/m2) for a sample of tablet computers. We want to know if screen brightness is...
-
Calculate how many milliliters of 0.626 M KOH should be added to 5.00 g of MOBS (Table 8-2) to give a pH of 7.40.
-
(a) Use Equations 8-20 and 8-21 to find the pH and concentrations of HA and A- in a solution prepared by mixing 0.00200 mol of acetic acid plus 0.004 00 mol of sodium acetate in 1.00 L of water. (b)...
-
(a) Calculate the pH of a solution prepared by mixing 0.0100 mol of the base B (Kb = 10 = - 2.00) with 0.020 0 mol of BH+Br- and diluting to 1.00 L. First calculate the pH by assuming [B] = 0.0100...
-
What are the potential consequences of neglecting or underestimating the importance of Risk and Threat Assessment in security and how or if it does influence of crime and criminology in your...
-
A runner maintains constant acceleration after starting from rest as she runs a distance of 60.0 m. The runner's speed at the end of the 60.0 m is 9.00 m/s. How much time did it take the runner to...
-
7. A ball is projected upward at time t = 0.0 s, from a point on a roof 60 m above the ground. The ball rises, then falls and strikes the ground. The initial velocity of the ball is 71.6 m/s if air...

Study smarter with the SolutionInn App


