A chemical reaction takes place in a series of four continuous stirred tank reactors arranged as shown
Question:
A chemical reaction takes place in a series of four continuous stirred tank reactors arranged as shown in Figure E 23.
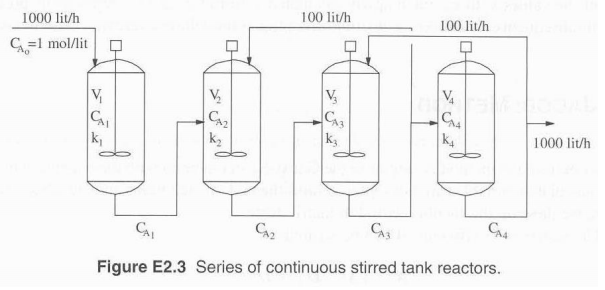
The chemical reaction is a first-order irreversible reaction of the type ![]()
The conditions of temperature in each reactor are such that the value of the rate constant k i , is different in each reactor. Also, the volume of each reactor V i , is different. The values of k i and V i are given in Table E 2.3. The following assumptions can be made regarding this system:
1. The system is at steady state.
2. The reactions are in the liquid phase.
3. There is no change in volume or density of the liquid.
4. The rate of disappearance of component A in each reactor is given by
R i = V i K i C Ai mol/h

Set up the material balance equation for each of the four reactors. What type of equations do you have in this set of material balances?





