An anti-anxiety medication is tested in a clinical trial using a factorial design. Specifically, 20 participants were
Question:
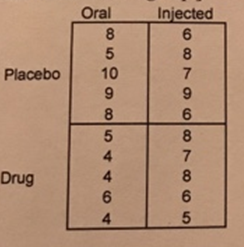
An anti-anxiety medication is tested in a clinical trial using a factorial design. Specifically, 20 participants were randomly assigned to receive either active drug or placebo. These two conditions were further divided into two drug administration methods. Specifically, half of the participants in each drug condition administered the medication orally (each day) whereas the other half was given a one-time long-lasting injection at the beginning of the study. The researchers predicted (a) that individuals getting the active drug, versus those taking placebo, would have lower anxiety, (b) individuals taking the drug orally, versus those getting the injection, will have lower anxiety, and (c) the effect of drug on reducing anxiety would be stronger in the oral administration condition compared to the long-lasting injection condition. Below are the data for the four groups. Evaluate the predictions (using an alpha level of .05). Provide SPSS output (with explanation of the findings) and a bar chart showing the pattern of means. [Note, the values in each box correspond to the 5 anxiety scores for the individuals in each group.]

Probability and Statistics
ISBN: 978-0321500465
4th edition
Authors: Morris H. DeGroot, Mark J. Schervish




