The following data for water may or may not be useful for this quiz: You have a
Question:
The following data for water may or may not be useful for this quiz:
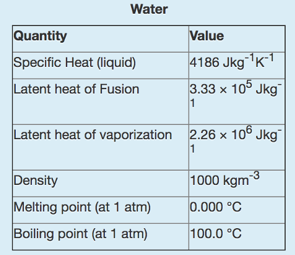
You have a metal block with a mass of 177.1 g, specific heat of 325 Jkg -1 k -1 , the thermal conductivity of 199.7 Wm -1 K -1 , an emissivity of 0.900 in air and an initial temperature of 81.7?C. The block is shaped as a square prism with a length of 5.66 cm and a cross-sectional area of 10.0 cm 2 .(that is the area of the square sides are 10.0 m 2 .) You start by dropping this metal block into an insulted 0.524 L bucket of water. The water is initially at 7.80?C.
What is the final temperature of the water?
T F = ??C
You now take the metal block and return it to its initial temperature before placing it in a room at a temperature of 14.1?C. What is the rate at this block initially loses energy? Give your answer a positive number as the block lose energy. You may assume that conservation is not important, heat is lost due to radiation.
P = ?W
What temperature would block finally reach if left in the room for a very long time?
T F = ??C
The block is now placed between a heat reservoir at 75.0?C and cold reservoir at 5.94?C (heat reservoirs are kept a constant temperature, either through them or cooling them). When the system has reached state of equilibrium what is the rate of heat flow along the block? The square sides of the block are up against the heat reservoirs.
P = ?W.

Physics
ISBN: 978-0077339685
2nd edition
Authors: Alan Giambattista, Betty Richardson, Robert Richardson




