The ideal gas law provides one way to estimate the pressure exerted by a gas in a
Question:
The ideal gas law provides one way to estimate the pressure exerted by a gas in a container. The law is
P = nRT/V
More accurate estimates can be made with the van der Waals equation:
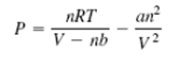
where the term nb is a correction for the volume of the molecules, and the term an 2 /V 2 is a correction for molecular attractions for molecular attractions. The values of a and b depend on the type of gas. The gas constant is R, the absolute temperature is T, the gas volume is V, and the number of gas molecules is indicated by n. If n = 1 mol of an ideal gas were confined to a volume of V = 22.41 L at 0?C (273.2 K), it would exert a pressure of 1 atmosphere. In these units, R = 0.08206.
For chlorine (Cl 2 ), a = 6.49 and b = 0.0562. Compare the pressure estimates given by the ideal gas law and the van der Waals equation for 1 mol of Cl 2 in 22.41 L at 273.2 K. What is the main cause of the difference in the two pressure estimates: the molecular volume or the molecular attractions?





