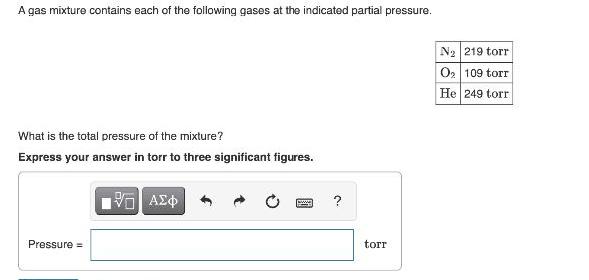
A gas mixture contains each of the following gases at the indicated partial pressure. N2 219...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A gas mixture contains each of the following gases at the indicated partial pressure. N2 219 torr O2 109 torr He 249 torr What is the total pressure of the mixture? Express your answer in torr to three significant figures. Pressure = torr A gas mixture contains each of the following gases at the indicated partial pressure. N2 219 torr O2 109 torr He 249 torr What is the total pressure of the mixture? Express your answer in torr to three significant figures. Pressure = torr
Expert Answer:
Answer rating: 100% (QA)
Any Axcording lo Daltons law q Accon 9 parlial presuve... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these chemistry questions
-
A gas mixture contains 10.0 mole% H 2 O (v) and 90.0 mole% N 2 . The gas temperature and absolute pressure at the start of each of the three parts of this problem are 50C and 500 mm Hg. Ideal gas...
-
A gas mixture contains 5 kg of N2 and 10 kg of O2. Determine The average molar mass and Gas constant.
-
Answer the questions about each of the following items related to a companys activities for the year: a. After using an expected useful life of seven years and no residual value to depreciate its...
-
What responsibilities can a Crew Boss delegate to a subordinate supervisor? (Select all that apply) Re-supplying crew and equipment Communicating crew wake up time for next operational period...
-
True or False: When the interest rate falls, people want to borrow more and the additional borrowing tends to drive the interest rate back up.
-
Discuss the notion of interviewer cheating. Why do interviewers cheat? How can cheating be detected and prevented?
-
Wash alumina solids to remove \(\mathrm{NaOH}\) from the entrained liquid. Underflow from the settler tank is \(20.0 \mathrm{vol} \%\) solid and \(80.0 \mathrm{vol} \%\) liquid. Two feeds to the...
-
Refer to Exercise 6-1. Calculate the amount of the noncontrolling interest to be deducted from consolidated income in arriving at 2011 controlling interest in consolidated net income.
-
Describe the marshmallow challenge and how does it relate to teams. When given a task like the marshmallow challenge, what do most teams do? Why are kindergartners better at the marshmallow challenge...
-
Congratulations! You've just been hired by a company as their new Quality Manager. They are a large manufacturer of parts for the aerospace industry. The boss sits you down on the first day and...
-
A market research team compiled the following discrete probability distribution for families residing in Randolph County. In this distribution,x represents the number of evenings the family dines...
-
Assuming the user enters a and b as positive integers. The output of the program is a= int(input()) b= int(input()) q=a//b r=a%b if a ==(q*b+r): print("hi") else: print("bye")
-
Stanley-Morgan Industries adopted a defined benefit pension plan on April 12, 2024. The provisions of the plan were not made retroactive to prior years. A local bank, engaged as trustee for the plan...
-
what surprised you about the future trends and supply of healthcare workers? Why might the local supply of healthcare professionals look very different than the state or national supply? Which types...
-
On January 2 of the current year, Black acquired a 5 0 % interest in New Partnership by contributing property with an adjusted basis of $ 7 , 0 0 0 and a fair market value of $ 9 , 0 0 0 , subject to...
-
Craftmore Machining reports the following budgeted overhead cost and related data for this year. Activity Assembly Product design Electricity Setup Total Budgeted Cost $ 438,750 Activity Cost Driver...
-
What is the intended purpose of this passage? To show the difference between ivory and plastics. To educate readers about how ivory and terrorism are connected. To describe why ivory products are so...
-
A crop-dusting plane flies over a level field at a height of 25 ft. If the dust leaves the plane through a 30 angle and hits the ground after the plane travels 75 ft, how wide a strip is dusted? See...
-
A liquid mixture containing 70.0 mol of n -pentane and 30.0 mol of n -hexane initially at 46?C is partially vaporized at P = 1 atm in a single-stage distillation apparatus (Rayleigh still). The heat...
-
Vapor pressure data for chlorine are given below. a. Use these data and the ClausiusClapeyron equation (Equation 6.1-3) to estimate the heat of vaporization of chlorine (kJ/mol) and to obtain an...
-
The CPVC wetcake leaves the product centrifuge at 80C and 1 atm and is fed to the elevated end of a rotary dryer (a rotating cylinder inclined about 35 from horizontal). A fresh air stream at 27C, 1...
-
The following transactions occurred during January 2019 for Richards Manufacturing Company: Jan. 5 Acquired $4,500 of materials on account that will be used to produce product for resale. 11...
-
Lederman Manufacturing Corporation sold 14,000 units of product for $45 each during 2019. During the year, 5,000 shares of common stock were outstanding. Prepare an income statement for the year...
-
Record the following transactions that occurred during March 2019 for Harris Manufacturing Company, which uses the perpetual inventory system: Mar. 12 Transferred $5,000 of completed goods from the...

Study smarter with the SolutionInn App


