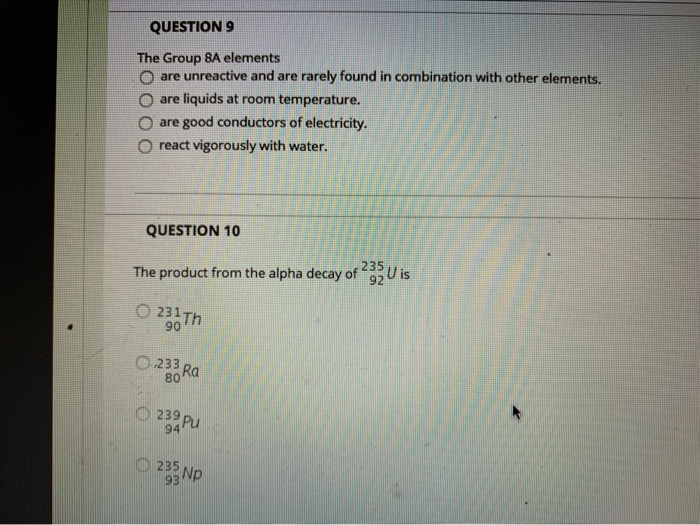
QUESTION 9 The Group 8A elements Oare unreactive and are rarely found in combination with other...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
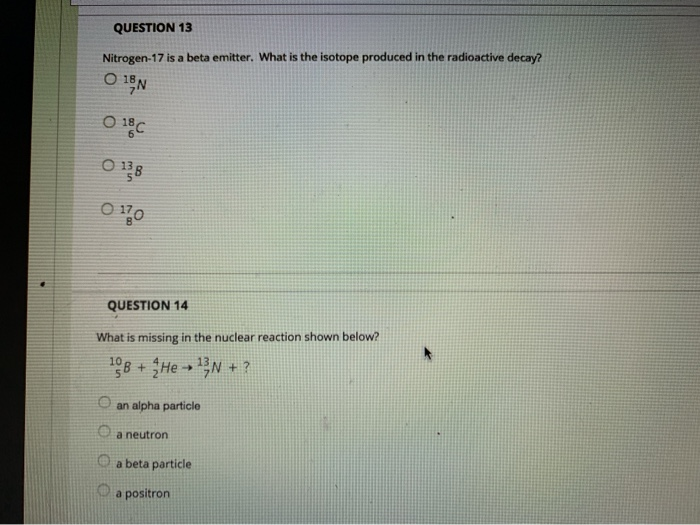
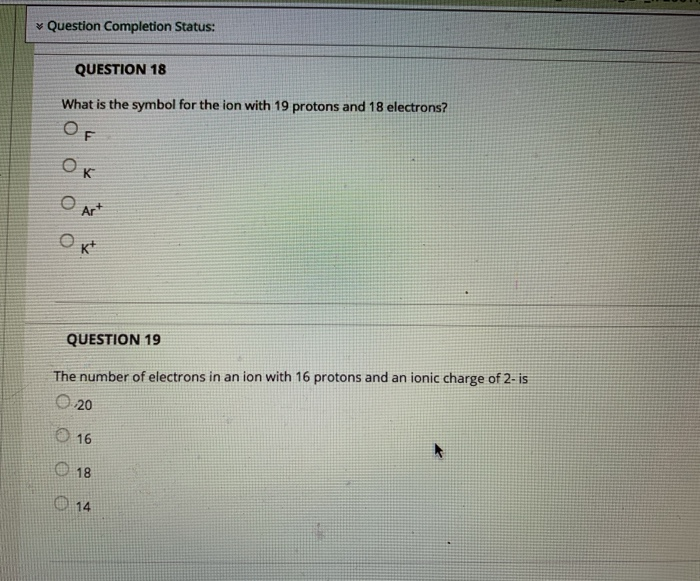
QUESTION 9 The Group 8A elements Oare unreactive and are rarely found in combination with other elements. are liquids at room temperature. are good conductors of electricity. react vigorously with water. QUESTION 10 The product from the alpha decay of O231 Th 90 O233 80 Ra 23⁹ Pu 94 235 93 Np 235 92 U is QUESTION 11 The process in which a nucleus spontaneously breaks down by emitting radiation is known as O a chain reaction O radioactive decay O fusion O fission QUESTION 12 Which is NOT a way to minimize your exposure to radiation? Ostaying a longer time 'wearing a lead apron waring lead-lined gloves keeping a good distance QUESTION 13 Nitrogen-17 is a beta emitter. What is the isotope produced in the radioactive decay? O BN 0 180 0 138 0170 QUESTION 14 What is missing in the nuclear reaction shown below? ¹8+He¹N+? O an alpha particle a neutron a beta particle a positron QUESTION 15 The octet rule indicates that O atoms lose, gain, or share valence electrons to have 8 valence electrons. all of the shells in an atom hold a maximum of 8 electrons. the noble gases react with other compounds to get 8 valence electrons. all of the noble gases have eight total electrons. QUESTION 16 In ionic compounds, O metals; cations metals; anions nonmetals; anions 'metals; polyatomic ions lose their valence electrons to form positively charged. Question Completion Status: QUESTION 18 What is the symbol for the ion with 19 protons and 18 electrons? OF OK O Art Okt QUESTION 19 The number of electrons in an ion with 16 protons and an ionic charge of 2- is 020 16 18 14 QUESTION 9 The Group 8A elements Oare unreactive and are rarely found in combination with other elements. are liquids at room temperature. are good conductors of electricity. react vigorously with water. QUESTION 10 The product from the alpha decay of O231 Th 90 O233 80 Ra 23⁹ Pu 94 235 93 Np 235 92 U is QUESTION 11 The process in which a nucleus spontaneously breaks down by emitting radiation is known as O a chain reaction O radioactive decay O fusion O fission QUESTION 12 Which is NOT a way to minimize your exposure to radiation? Ostaying a longer time 'wearing a lead apron waring lead-lined gloves keeping a good distance QUESTION 13 Nitrogen-17 is a beta emitter. What is the isotope produced in the radioactive decay? O BN 0 180 0 138 0170 QUESTION 14 What is missing in the nuclear reaction shown below? ¹8+He¹N+? O an alpha particle a neutron a beta particle a positron QUESTION 15 The octet rule indicates that O atoms lose, gain, or share valence electrons to have 8 valence electrons. all of the shells in an atom hold a maximum of 8 electrons. the noble gases react with other compounds to get 8 valence electrons. all of the noble gases have eight total electrons. QUESTION 16 In ionic compounds, O metals; cations metals; anions nonmetals; anions 'metals; polyatomic ions lose their valence electrons to form positively charged. Question Completion Status: QUESTION 18 What is the symbol for the ion with 19 protons and 18 electrons? OF OK O Art Okt QUESTION 19 The number of electrons in an ion with 16 protons and an ionic charge of 2- is 020 16 18 14
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below 9The group 8 A elements unreactive and rarely found in combination with other elements Th... View the full answer

Related Book For 

A Concise Introduction to Logic
ISBN: 978-1305958098
13th edition
Authors: Patrick J. Hurley, Lori Watson
Posted Date:
Students also viewed these chemistry questions
-
Good conductors of electricity are often good conductors of heat. Why might this be?
-
Until the early 1960s the group 8A elements were called the inert gases; before that they were called the rare gases. The term rare gases were dropped after it was discovered that argon accounts for...
-
A 236U nucleus undergoes fission and breaks into two middle- mass fragments, 140Xe and 96Sr. (a) By what percentage does the surface area of the fission products differ from that of the original 236U...
-
The cantilever beam AB shown in the figure is subjected to a concentrated load P at the midpoint and a counterclockwise couple of moment M1 = PL/4 at the free end. Draw the shear-force and...
-
The following is an excerpt from a conversation between two sales clerks, Fred Loya and Steph Gillespie. Both Fred and Steph are employed by Wireless Electronics, a locally owned and operated...
-
Using, Exhibit 13-12, the balance sheet of Angelinas Jewelry Company at the end of July shown below, calculate all four financial ratios (quick, current, debt, and debt-to-equity) for the business....
-
Let \(f(x)\) be defined for \(x \in[-L, L]\). Parseval's identity is given by \[\frac{1}{L} \int_{-L}^{L} f^{2}(x) d x=\frac{a_{0}^{2}}{2}+\sum_{n=1}^{\infty} a_{n}^{2}+b_{n}^{2}\] Assuming the the...
-
In an attempt to judge and monitor the quality of instruction, the administration of Mega- Byte Academy devised an examination to test students on the basic concepts that all should have learned....
-
Discuss in thr..ee paragr.aphs Why corporations are advised to buy liability insurance? 1. You should wri.te si.x..hund.red wo.rds at least. 2. The report should be wri..tten in your own language .
-
During the current year, Yost Company disposed of three different assets. On January 1 of the current year, prior to the disposal of the assets, the accounts reflected the following: The machines...
-
Charismatic Leadership. Discuss your not in agreement with the the Charismatic leadership role, using academic journals to support your position. What are the pros and cons of that. Using 250 words...
-
On January 1, Carla Vista Corporation purchased a 25% equity investment in Shane Corporation for $140,300. At December 31, Shane declared and paid a $80,600 cash dividend and reported net income of...
-
In 2022, Alliant Corporation acquired Centerpoint Incorporated for $339 million, of which $59 million was allocated to goodwill. At the end of 2024, management has provided the following information...
-
In 1 9 5 2 Blayney Scott and his wife Almeda started a small company in Victoria, British Columbia, Canada that pioneered the use of plastics in the manufacturing of marine products. Scotty...
-
Land Co wanted to purchase a plot of land. They agreed to purchase a plot of land with a building on it with the intention of only keeping the land. The assessment value of the land by an independent...
-
I need help with following parts relating to this question: 1. The manufacturer has collected monthly data on past market prices of widgets. Suppose that all annual price change can be assumed to be...
-
Rodger Corporation's 12% coupon rate, semiannual payment, $1,000 par value bonds, which mature in 25 years, are callable 6 years from today at $1,025. They sell at a price of $1,278.56, and the yield...
-
Transform the while loop from the previous exercise into an equivalent for loop (make sure it produces the same output).
-
(K H) (H K) Use truth tables to determine whether the following symbolized statements are tautologous, self-contradictory, or contingent.
-
Exercises 11 through 20 provide a statement, its truth value in parentheses, and a new statement. Determine how the new statement was derived from the given statement and supply the truth value of...
-
1. If government deficits continue at their present rate and a recession sets in, then interest on the national debt will become unbearable and the government will default on its loans. If a...
-
On December 31, 2020, for GAAP purposes, Clubs Inc. reported a balance of \(\$ 70,000\) in Prepaid Maintenance Expense for services to be received over the following year. For tax purposes, however,...
-
On December 31, 2020, for GAAP purposes, Clubs Inc. reported a balance of \(\$ 40,000\) in a warranty liability for anticipated costs to satisfy future warranty claims. No claims were paid in 2020....
-
The records of Anderson Inc. provide the following information for the tax year 2020. - There was no beginning balance in deferred tax account(s). - Taxable income for 2020 was \(\$ 60,000\). - Tax...

Study smarter with the SolutionInn App


