SOLVE THE FOLLOWING Sheet of current: Hoi Then figure out the following limiting cases: Consider an
Fantastic news! We've Found the answer you've been seeking!
Question:
SOLVE THE FOLLOWING
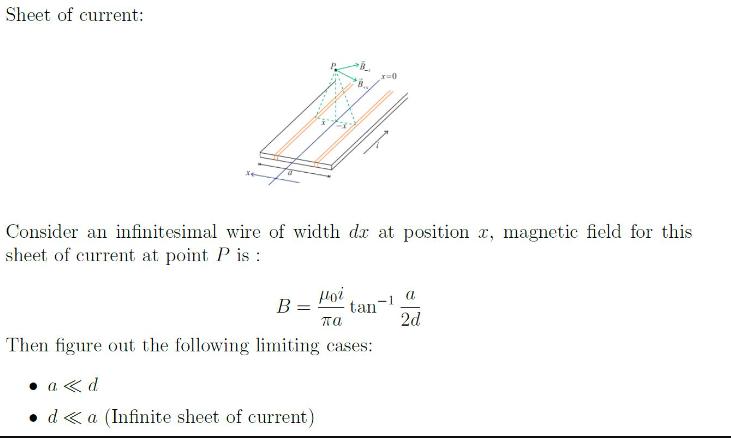
Transcribed Image Text:
Sheet of current: Hoi πα Then figure out the following limiting cases: Consider an infinitesimal wire of width de at position a, magnetic field for this sheet of current at point P is : B = tan x=0 a<<<d • d <a (Infinite sheet of current) - -1 a 2d Sheet of current: Hoi πα Then figure out the following limiting cases: Consider an infinitesimal wire of width de at position a, magnetic field for this sheet of current at point P is : B = tan x=0 a<<<d • d <a (Infinite sheet of current) - -1 a 2d
Expert Answer:
Answer rating: 100% (QA)
Current sheets in plasmas store energy by increasing the energy density of the magnetic field Many p... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
0.50 mole of CH3Br and 0.75 mole of H2S are injected into a 1.00 L reaction vessel at 500 K. CH;Br(g) + H2S(g) CH,SH(g) + HBr(g) K, for the reaction at 500 K is 0.33. What are the equilibrium...
-
A reaction vessel contains NH3, N2, and H2 at equilibrium at a certain temperature. The equilibrium concentrations are [NH3] = 0.25 M, [N2] = 0.11 M, and [H2] = 1.91 M. Calculate the equilibrium...
-
At 273 K and 1 atm pressure, one mole of an ideal gas occupies (a) Looking back at Figure 18.1, do you predict that 1 mole of an ideal gas in the middle of the stratosphere would occupy a greater or...
-
Consider a soap bubble. Is the pressure inside the bubble higher or lower than the pressure outside?
-
List the advantages and disadvantages of a functional versus a divisional organizational structure.
-
Writing Improvement Exercise: Removing Fillers Your Task. Revise the following sentences to avoid fillers. a. There are many businesses that are considering strict e-mail policies. b. It is the CEO...
-
In 1990, Spirit Airlines began as a small, low-fare passenger airline. By 1996 it had experienced modest success and had grown to include 11 planes and about 455 employees. In 1995 and 1996, it...
-
Consider the following transactions for Huskies Insurance Company: a. Equipment costing $42,000 is purchased at the beginning of the year for cash. Depreciation on the equipment is $7,000 per year....
-
Automobile demand has fallen during a recent recession, and Ana has been laid off from her job on the assembly line. Rajiv recently lost his job as a waiter at a local restaurant. A recent increase...
-
It is October 16, 2020, and you have just taken over the accounting work of China Moon Products, whose annual accounting period ends October 31. The company?s previous accountant journalized its...
-
Write your initials on a piece of paper and take their pictures. (a) Plot each letter with its corresponding 2D Fourier transform. Use characters from any language you wish. (You can also see some...
-
At a temperature of 60.6C, benzene exerts a saturation pressure of 400 torr. At 80.1C, its saturation pressure is 760 torr. Using only these data, estimate the enthalpy of vaporization of benzene....
-
Reread this chapters opening case. What factors did Sunil Rajaraman have working in his favor, and what factors did he have working against him as the cofounder of Scripped?
-
At 922 K, the enthalpy of liquid Mg is 26.780 [kJ/mol] and the entropy is 73.888 [J/(mol K)]. Determine the Gibbs energy of liquid Mg at 1300 K. The heat capacity of the liquid is constant over this...
-
What are the important changes that have occurred in stock markets since 2005?
-
The Nelson partnership begins business on July 17, 2018. It is not able to use business purpose to support a tax year. The four partners, their interests in partnership capital and profits, and their...
-
The theory that best advocates for a board made up of a majority of non-independent directors is: QUESTION 7SELECT ONE: a. Shareholder theory b. Stewardship Theory c. Stakeholder Theory (managerial...
-
The landing gear of an aircraft with: mass of 2000 kg the spring-mass-damper system Consider that the runway surface is y(t) = 0.2 cos 157.08t stiffness of the spring is 5 x 105 N/m. What is the...
-
The HR (human resources) department has requested some assistance. They would like to identify all employees who are not withholding the maximum savings amount for the 401K retirement plan of 6% to...
-
How much cash did Mr. Ames receive from the Soviet Union for spying?
-
Why is the Internet such an attractive operational location for criminal enterprises? 1. Criminals can profit by stealing and selling information. 2. Cyberspace gives criminals a worldwide reach. 3....

Study smarter with the SolutionInn App

