25.00mL of Sodium Hydroxide solution was titrated with 23.40mL of 0.5720N Sulphuric Acid. Find the concentration...
Fantastic news! We've Found the answer you've been seeking!
Question:
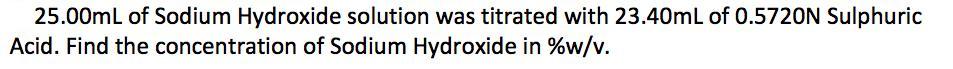
Transcribed Image Text:
25.00mL of Sodium Hydroxide solution was titrated with 23.40mL of 0.5720N Sulphuric Acid. Find the concentration of Sodium Hydroxide in %w/v. 25.00mL of Sodium Hydroxide solution was titrated with 23.40mL of 0.5720N Sulphuric Acid. Find the concentration of Sodium Hydroxide in %w/v.
Expert Answer:
Posted Date:
Students also viewed these chemistry questions
-
Question 2 Refer to the following figure. ID = 6 mA. The value of VDS is equal to------. Not yet +12 V answered 1 kn Marked out of 1.00 P Flag question 10 Ma Select one: O a. 6 V b. -6 V O c. -3 V O...
-
Determine the volume of sodium hydroxide solution needed to prepare 26.2 g sodium phosphate, Na3PO4, by the reaction 3NaOH(aq) + H3PO4(aq) Na3PO4(aq) + 3H2O(l) The sodium hydroxide solution, whose...
-
A. Find the concentration of salt (lb/gal) in tank 1 and 2 after 100 minutes. B. Find the concentration of salt (lb/gal) in tank 1 and 2 when tank 2 is in full capacity. C. Find the concentration of...
-
In Problem 10.16, we projected financial statements for Walmart Stores for Years +1 through +5. The data in Chapter 12s Exhibits 12.1612.18 include the actual amounts for 2008 and the projected...
-
In what signicant ways would each of the three major statements of a government hospital differ from those of a private not-for-prot hospital?
-
A large manufacturing company produces computer printers that are distributed and sold all over the United States. Due to lack of industry information, the company has a difficult time ascertaining...
-
Your classmate, Joe Livingston, is confused about the factors that are included in the annual rate of return technique. What is the formula for this technique?
-
Calm Day reported the following income statement for the year ended December 31, 2019: Requirements 1. Compute Calm Day's inventory turnover rate for the year. (Round to two decimal places.) 2....
-
Circle B is a dilation of circle A. circle A 3 1. What is the scale factor of dilation? circle B 2. What is the length of the highlighted arc in circle A? 6 units 3. What is the length of the...
-
How could the owners of Willow Springs have ensured the safety of their customers and prevented the infections from occurring? Explain.
-
Lt's pretend that our company is TARGET stores. a. Apply the Porter's 5 forces model to the industry in which our company is based. What does this model tell you about the nature of competition in...
-
_____ consist of any action such as firings, demotions, schedule reductions, or changes that would harm the individual employee.
-
_____ measures a persons intellectual curiosity and motivation in a particular field.
-
_____ is a plan of action to achieve a particular set of objectives.
-
_____ is a notice from the EEOC, if they elect not to prosecute an individual discrimination complaint within the agency, that gives the recipient the right to go directly to the courts with a...
-
_____ consist of monetary damages awarded by the court that are designed to punish an injuring party that intentionally inflicted harm on others.
-
Blossom Ltd. had the following share transactions during its first year of operations: Jan. 6 Issued 240,000 common shares for $1.50 per share. Jan. 12 Issued 60,000 common shares for $1.75 per...
-
Why is inventory management important for merchandising and manufacturing firms and what are the main tradeoffs for firms in managing their inventory?
-
The Welding Department of Nagano Manufacturing Company has the following production and manufacturing cost data for February 2002. All materials are added at the beginning of the process. Nagano uses...
-
With the popularity of the Apple iPad, the tablet computer market is experiencing high growth rates and record demand levels. Using the Internet as a research tool, complete a five forces analysis...
-
The ledger of Grogan Company has the following work in process account. Production records show that there were 800 units in the beginning inventory, 30% complete, 1,100 units started, and 1,300...

Study smarter with the SolutionInn App


