1. (40 pts) Citric acid (C6H8O7) is used in the preparation of many foods, pharmaceuticals, and...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
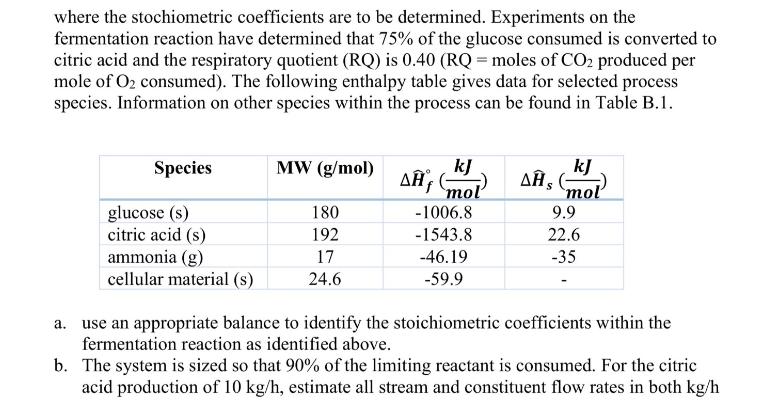
1. (40 pts) Citric acid (C6H8O7) is used in the preparation of many foods, pharmaceuticals, and personal care products. Although it can be recovered by concentration and crystallization from citrus juices, modern commercial production involves synthesis by fermentation of carbohydrates by Aspergillus niger. The process involves addition of the fungus to a fermenter along with carbohydrates, nutrients, water, and air that is bubbled through the fermentation broth. After the desired production of citric acid, the resultant fermentation liquor is filtered to remove the cell mass and other suspended solids followed by recovery and purification of citric acid by crystallization. As part of the evaluation of a proposed continuous fermentation process, you have been tasked with estimation of the required heating/cooling requirements for the fermenter designed to produce 10 kg/h of citric acid. Feed into the unit includes an aqueous solution that is 20 wt.% glucose, 0.4wt% ammonia, and the balance water. Air at 1.2 atm, saturated with water, is bubbled through fermenter providing a molar flowrate of oxygen twice that of glucose. One major stream leaving the fermenter is a gas stream at 1 atm containing nitrogen, the unreacted oxygen, carbon dioxide produced during the fermentation process. This gas stream is also saturated with water. The other major stream leaving the fermenter is a liquid fermentation liquor containing water, citric acid, unreacted glucose, and unreacted ammonia. All streams are assumed to exist at 25 C and the contents of the fermenter can be assumed to be well mixed due to the nature of air bubbling within the fermenter. The fermentation reaction is given as: C6H12O6 (aq) + a NH3(aq) + b O(9) C CH1.79N0.200.5 (s) + d CO2 + e HO (1) + f C6H8O7 (aq) where the stochiometric coefficients are to be determined. Experiments on the fermentation reaction have determined that 75% of the glucose consumed is converted to citric acid and the respiratory quotient (RQ) is 0.40 (RQ = moles of CO2 produced per mole of O2 consumed). The following enthalpy table gives data for selected process species. Information on other species within the process can be found in Table B.1. Species MW (g/mol) kJ kJ A mol , (mol glucose (s) citric acid (s) 180 -1006.8 9.9 192 -1543.8 22.6 ammonia (g) 17 -46.19 -35 cellular material (s) 24.6 -59.9 a. use an appropriate balance to identify the stoichiometric coefficients within the fermentation reaction as identified above. b. The system is sized so that 90% of the limiting reactant is consumed. For the citric acid production of 10 kg/h, estimate all stream and constituent flow rates in both kg/h and mol/h. What are the volumetric flow rates of air entering the fermenter and the off-gas stream (in L/h)? c. The heats of formation provided within the table indicate the phases of materials; however, the reaction is specified with substances within the aqueous phase. Show how Hess's law can be used to estimate the heat of formation in the aqueous phase based on the provided enthalpies for the gaseous and solid species. d. Estimate the rate at which heat must be added or removed from the fermenter (in kW). 1. (40 pts) Citric acid (C6H8O7) is used in the preparation of many foods, pharmaceuticals, and personal care products. Although it can be recovered by concentration and crystallization from citrus juices, modern commercial production involves synthesis by fermentation of carbohydrates by Aspergillus niger. The process involves addition of the fungus to a fermenter along with carbohydrates, nutrients, water, and air that is bubbled through the fermentation broth. After the desired production of citric acid, the resultant fermentation liquor is filtered to remove the cell mass and other suspended solids followed by recovery and purification of citric acid by crystallization. As part of the evaluation of a proposed continuous fermentation process, you have been tasked with estimation of the required heating/cooling requirements for the fermenter designed to produce 10 kg/h of citric acid. Feed into the unit includes an aqueous solution that is 20 wt.% glucose, 0.4wt% ammonia, and the balance water. Air at 1.2 atm, saturated with water, is bubbled through fermenter providing a molar flowrate of oxygen twice that of glucose. One major stream leaving the fermenter is a gas stream at 1 atm containing nitrogen, the unreacted oxygen, carbon dioxide produced during the fermentation process. This gas stream is also saturated with water. The other major stream leaving the fermenter is a liquid fermentation liquor containing water, citric acid, unreacted glucose, and unreacted ammonia. All streams are assumed to exist at 25 C and the contents of the fermenter can be assumed to be well mixed due to the nature of air bubbling within the fermenter. The fermentation reaction is given as: C6H12O6 (aq) + a NH3(aq) + b O(9) C CH1.79N0.200.5 (s) + d CO2 + e HO (1) + f C6H8O7 (aq) where the stochiometric coefficients are to be determined. Experiments on the fermentation reaction have determined that 75% of the glucose consumed is converted to citric acid and the respiratory quotient (RQ) is 0.40 (RQ = moles of CO2 produced per mole of O2 consumed). The following enthalpy table gives data for selected process species. Information on other species within the process can be found in Table B.1. Species MW (g/mol) kJ kJ A mol , (mol glucose (s) citric acid (s) 180 -1006.8 9.9 192 -1543.8 22.6 ammonia (g) 17 -46.19 -35 cellular material (s) 24.6 -59.9 a. use an appropriate balance to identify the stoichiometric coefficients within the fermentation reaction as identified above. b. The system is sized so that 90% of the limiting reactant is consumed. For the citric acid production of 10 kg/h, estimate all stream and constituent flow rates in both kg/h and mol/h. What are the volumetric flow rates of air entering the fermenter and the off-gas stream (in L/h)? c. The heats of formation provided within the table indicate the phases of materials; however, the reaction is specified with substances within the aqueous phase. Show how Hess's law can be used to estimate the heat of formation in the aqueous phase based on the provided enthalpies for the gaseous and solid species. d. Estimate the rate at which heat must be added or removed from the fermenter (in kW).
Expert Answer:

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, ? Ronald W. Rousseau, ? Lisa G. Bullard
Posted Date:
Students also viewed these chemical engineering questions
-
Citric acid (C 6 H 8 O 7 ) is used in the preparation of many foods, pharmaceuticals, soft drinks, and personal-care products. Although it can be recovered by concentration and crystallization from...
-
Q1. You have identified a market opportunity for home media players that would cater for older members of the population. Many older people have difficulty in understanding the operating principles...
-
Uniform rod AB of length l and mass m lies in a vertical plane and is acted upon by a couple M. The ends of the rod are connected to small rollers which rest against frictionless surfaces. (a)...
-
A machine purchased three years ago for $720,000 has a current book value using straight-line depreciation of $400,000; its operating expenses are $60,000 per year. A replacement machine would cost...
-
John Luis receives wages from three employers during 2017. Is he entitled to a refund on the OASDI taxes paid on wages in excess of the $118,500 limit? If so, how can he receive a refund
-
True or False. Vibration can cause structural and mechanical failures.
-
Swinn Company manufactures bicycles. Materials are added at the beginning of the production process, and conversion costs are incurred uniformly. Production and cost data for the month of May are as...
-
A manufacturer uses activity-based costing to assign overhead cost to products. Budgeted cost information for its activities follows Activity Purchasing Factory services Setup Budgeted Cost $ 175,000...
-
You interviewed well and got the job on Wall Street in NYC, a place that is notorious for working its new employees to extremes. Raises are given after 6 months, with the amount of time you put in...
-
A market structure characterized by many sellers with each having some pricing power and product differentiation is best described as: A. oligopoly. B. perfect competition. C. monopolistic...
-
A company doing business in a monopolistically competitive market will most likely maximize profits when its output quantity is set such that: A. average cost is minimized. B. marginal revenue equals...
-
Movement along the demand curve for good \(X\) occurs due to a change in: A. income. B. the price of \(\operatorname{good} X\). C. the price of a substitute for \(\operatorname{good} X\).
-
The numerator of the GDP price deflator reflects: A. the value of base-year output at current prices. B. the value of current-year output at current prices. C. the value of current-year output at...
-
In the case of two goods, \(x\) and \(y\), which of the following statements is most likely true? Maximum utility is achieved: A. along the highest indifference curve below the budget constraint...
-
Windsor Company purchased machinery for $595800 on January 2, 2023. Straight-line depreciation has been recorded based on a $30600 salvage value and a 3-year useful life. The machinery was sold on...
-
Problem 2. (0.6 points, 0.2 points for each question) (a) A company turns its inventory 2 times a month. Its months-of-supply = Its days-of-supply = Please show your analysis below: _months. days. (1...
-
Rain is falling on a poorly designed 150-m2 flat roof of a contemporary home. The design flaw requires the water level on the roof to reach approximately 5 cm above the plane of the roof before the...
-
Several decades ago benzene was thought to be a harmless chemical with a somewhat pleasant odor and was widely used as a cleaning solvent. It has since been found that chronic exposure to benzene can...
-
As described in Problem 9.21, the manufacture of ethanol from corn starch involves fermentation using a yeast that converts sugars from the starch to ethanol and carbon dioxide in a complicated...
-
Look up the following comovements for the country in which you are studying on an appropriate website. Explain whether the comovements are procyclical or countercyclical; GDP and: a. inflation b....
-
Which business cycle model do you find the most compelling and why?
-
To what extent do you think that workers are always fooled by the inflation fallacy?

Study smarter with the SolutionInn App


