1. a. Calculate the rate constant at the higher temperature used in Experiment 4. b. Calculate the...
Fantastic news! We've Found the answer you've been seeking!
Question:
1. a. Calculate the rate constant at the higher temperature used in Experiment 4.
b. Calculate the activtion energy for the reaction.
c. What do your results demotrate about the effect of a catalyst on the rate of reaction.

Transcribed Image Text:
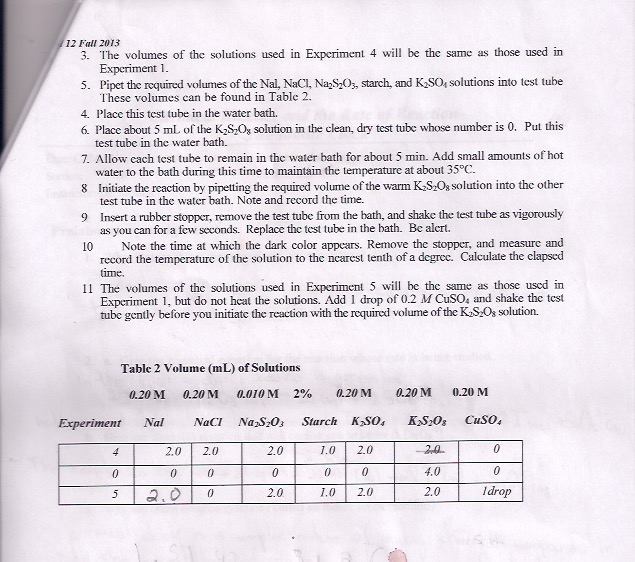
12 Fall 2013 3. The volumes of the solutions used in Experiment 4 will be the same as those used in Experiment 1. 5. Pipet the required volumes of the Nal, NaCl, Na₂S₂O3, starch, and K₂SO, solutions into test tube These volumes can be found in Table 2. 4. Place this test tube in the water bath. 6. Place about 5 mL of the K₂S₂Ox solution in the clean, dry test tube whose number is 0. Put this test tube in the water bath. 7. Allow each test tube to remain in the water bath for about 5 min. Add small amounts of hot water to the bath during this time to maintain the temperature at about 35°C. 8 Initiate the reaction by pipetting the required volume of the warm K₂S₂O, solution into the other test tube in the water bath. Note and record the time. 9 Insert a rubber stopper, remove the test tube from the bath, and shake the test tube as vigorously as you can for a few seconds. Replace the test tube in the bath. Be alert. 10 Note the time at which the dark color appears. Remove the stopper, and measure and record the temperature of the solution to the nearest tenth of a degree. Calculate the clapsed time. 11 The volumes of the solutions used in Experiment 5 will be the same as those used in Experiment 1, but do not heat the solutions. Add 1 drop of 0.2 M CuSO4 and shake the test tube gently before you initiate the reaction with the required volume of the K₂S₂Os solution. Table 2 Volume (mL) of Solutions 0.20 M 0.20 M Experiment Nal 4 0 5 2.0 0 NaCl 2.0 0 0 0.010 M 2% 0.20 M Na₂S₂O3 2.0 0 2.0 Starch K₂SO4 1.0 2.0 0 0 1.0 2.0 0.20 M K₂S208 -2,0 4.0 2.0 0.20 M CuSO4 0 0 Idrop 12 Fall 2013 3. The volumes of the solutions used in Experiment 4 will be the same as those used in Experiment 1. 5. Pipet the required volumes of the Nal, NaCl, Na₂S₂O3, starch, and K₂SO, solutions into test tube These volumes can be found in Table 2. 4. Place this test tube in the water bath. 6. Place about 5 mL of the K₂S₂Ox solution in the clean, dry test tube whose number is 0. Put this test tube in the water bath. 7. Allow each test tube to remain in the water bath for about 5 min. Add small amounts of hot water to the bath during this time to maintain the temperature at about 35°C. 8 Initiate the reaction by pipetting the required volume of the warm K₂S₂O, solution into the other test tube in the water bath. Note and record the time. 9 Insert a rubber stopper, remove the test tube from the bath, and shake the test tube as vigorously as you can for a few seconds. Replace the test tube in the bath. Be alert. 10 Note the time at which the dark color appears. Remove the stopper, and measure and record the temperature of the solution to the nearest tenth of a degree. Calculate the clapsed time. 11 The volumes of the solutions used in Experiment 5 will be the same as those used in Experiment 1, but do not heat the solutions. Add 1 drop of 0.2 M CuSO4 and shake the test tube gently before you initiate the reaction with the required volume of the K₂S₂Os solution. Table 2 Volume (mL) of Solutions 0.20 M 0.20 M Experiment Nal 4 0 5 2.0 0 NaCl 2.0 0 0 0.010 M 2% 0.20 M Na₂S₂O3 2.0 0 2.0 Starch K₂SO4 1.0 2.0 0 0 1.0 2.0 0.20 M K₂S208 -2,0 4.0 2.0 0.20 M CuSO4 0 0 Idrop
Expert Answer:
Answer rating: 100% (QA)
NaI moles in 2 mL 0221000 00004 moles persulphate moles in 2mL 0221000 00004 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the rate constant at 500 K for the second-order gas-phase reaction between Cl and H 2 , given the frequency factor, A-8.1x10 -10 dm 3 mol -1 s -1 and activation energy E a = 23 kJ mol.
-
In an experiment to determine the effect of catalyst on the yield of a certain reaction, the mean yields for reactions run with each of four catalysts were X1. = 89.88, X2. = 89.51, X3. = 86.98, and...
-
Calculate the energy change for the following nuclear reaction (in joules per mole of Give the energy change in MeV per nucleus. See Table 20.3. H) H +H Heon 97 97 98 98 01 02 03 03 04 03 04 04 OS...
-
Southern Stitches is a local casual clothing shop that makes a variety of t-shirt styles. Below is some actual vs. budget information for Southern's t-shirts for the month of May. Assume no...
-
Vons Grocery, a large retail grocery chain in Los Angeles, sought to acquire Shopping Bag Food Stores, a direct competitor. At the time of the proposed merger, Vons sales ranked third in the Los...
-
Identify the three major sections in the statement of cash flows, and briefly describe the nature of the items that appear in each section. Discuss.
-
If many operations have the same precedence in an expression, in what order should the operations be performed?
-
DeJohn Company, which began operations at the beginning of 2012, produces various products on a contract basis. Each contract generates a gross profit of $80,000. Some of DeJohns contracts provide...
-
5. In 2019, Susan makes the following donations to 50% qualified charitable organizations: Basis Fair Market Value Inventory held for resale in Susan's business (a sole proprietorship) $9,000 $...
-
Holly wants to explore the difference between buying and leasing a vehicle, both over 5 years with monthly payments. The car that she has an interest in has a value of $27000, and is located in...
-
A company expects sales of $1,500,000 next year and maintains an average inventory level of $250,000, accounts receivable of $300,000, and accounts payable of $200,000. Calculate the working capital...
-
Consider the following sequence and find the following (explain your ansv answers). (a) Plot the sequence y[n]? (b) Find the length of y[n]. (c) Size for e= 3? y[n] = [-n-2] - [n (d) Is this sequence...
-
. On a Mac, processes are listed on the CPU tab; on Windows, select the Processes tab. A process corresponds to a program. Any programs that are currently running put a load on the CPU. A. What is...
-
Bob received 5 cipher blocks Co. C1, C2, C3, C4 encrypted by CBC. When he tried to decrypt it to get Po, P1, P2, P3, P4, he accidentally used X instead of C2. Pick all the plaintext blocks that will...
-
Address the Pros and Cons pertaining to the viability, effectiveness, and fairness of each tax method? (must list 2 pros and cons for each) A. The governor is proposing a change to state's sales tax...
-
What was an effective part of the content of the speech? What was the most effective part of the delivery of the speech? How could content be improved? How could delivery be improved?
-
We learned about prejudice. One of the measures used in social psychology to measureunconscious (implicit) forms of prejudice is the Implicit Association Test (IAT). Your assignment for this week is...
-
What services are provided by the provincial and territorial governments?
-
In the presence of excess thiocyanate ion, SCN, the following reaction is first order in chromium(III) ion, Cr3+; the rate constant is 2.0 106/s. Cr3+(aq) + SCN(aq) Cr(SCN)2+(aq) If 85.0% reaction...
-
Ammonium sulfate is used as a nitrogen and sulfur fertilizer. It is produced by reacting ammonia with sulfuric acid. Write the balanced equation for the reaction of gaseous ammonia with sulfuric acid...
-
Explain why evaporation leads to cooling of the liquid.
-
The following are the attributes of an independent auditor, except: (a) To be independent an auditor must be without bias with respect to the client since otherwise, he would lack that impartiality...
-
In agreed-upon procedure, the specified parties are responsible for all, except: (a) Assume the risk that such procedures might be insufficient for their purposes (b) Assume the risk that they might...
-
Which of the following assertions is inaccurate may challenge the auditors independence in fact or/and the perceived independence? (a) An independent auditor auditing a company in which he was also a...

Study smarter with the SolutionInn App


