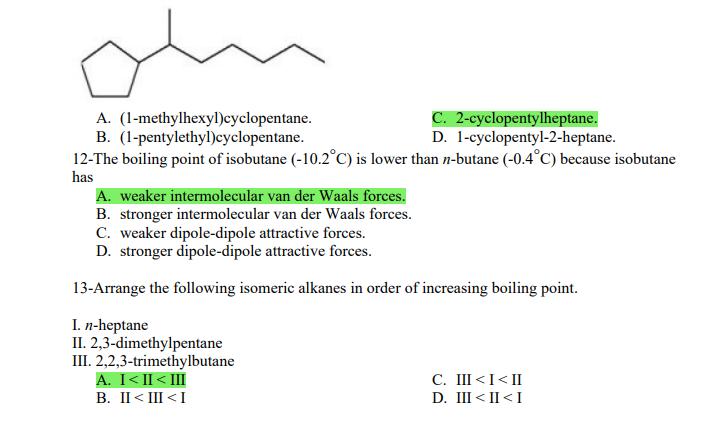
1- Alkanes are characterized by the general molecular formula: A. CnH2n-2 B. CnH2n C. CnH2n+2 2-Cycloalkanes...
Fantastic news! We've Found the answer you've been seeking!
Question:







Transcribed Image Text:
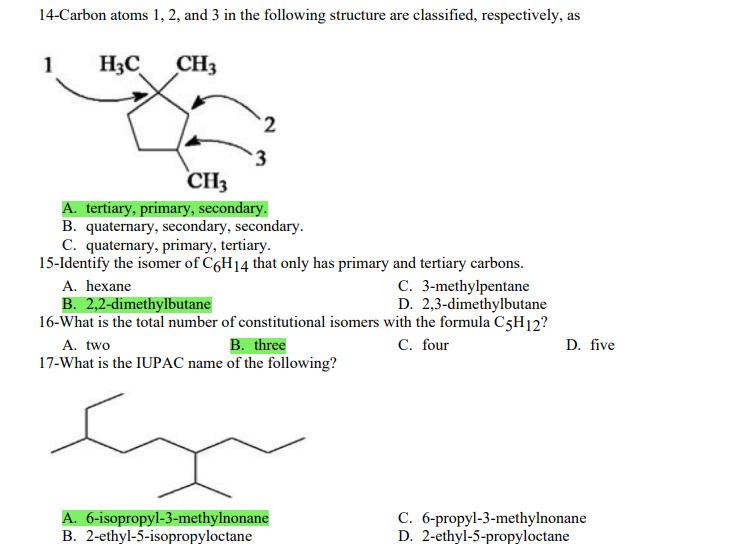
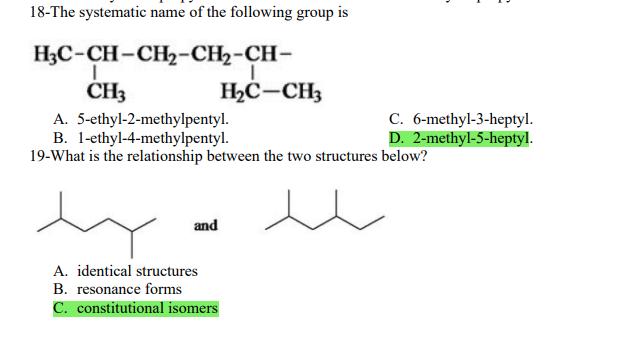
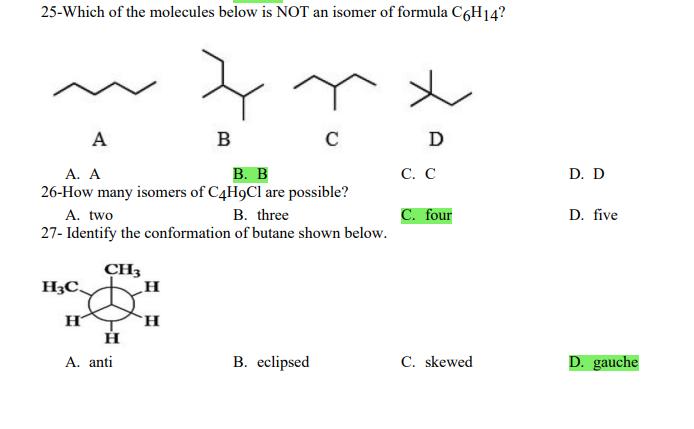
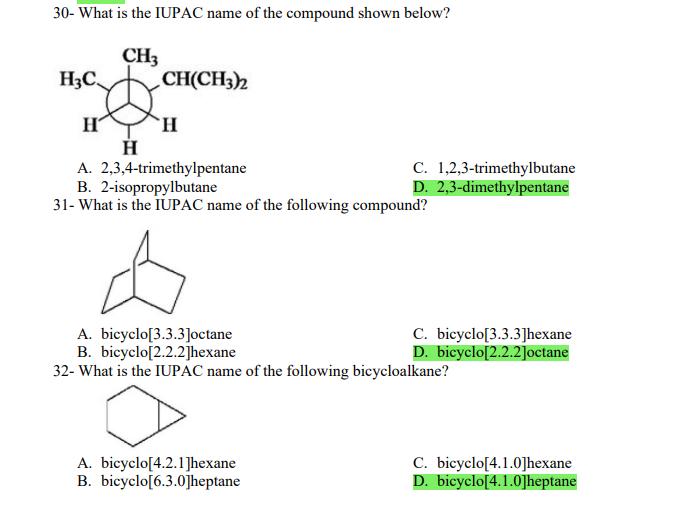
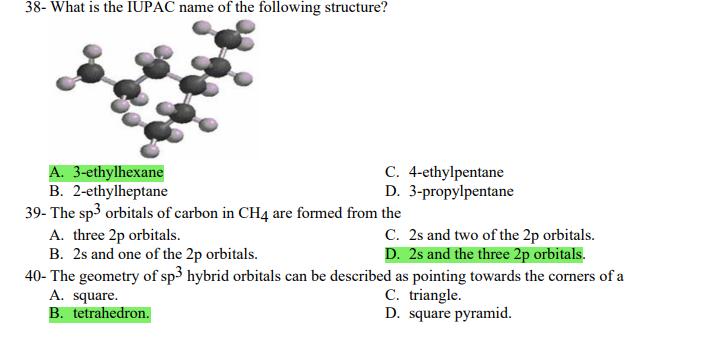
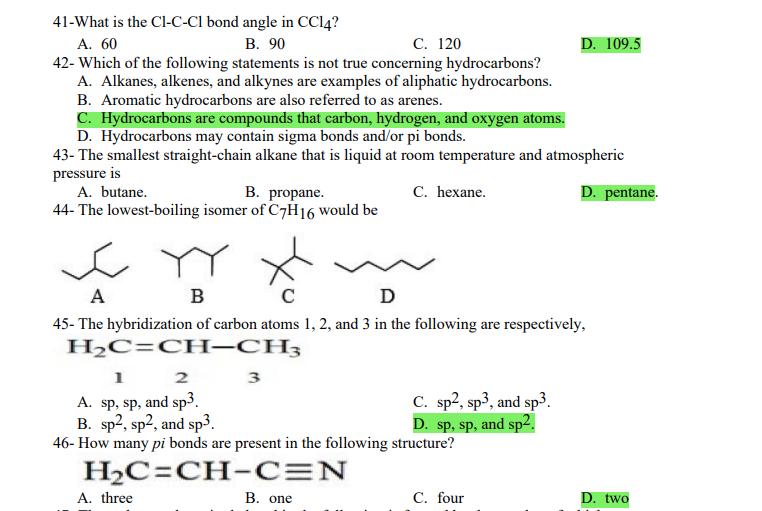
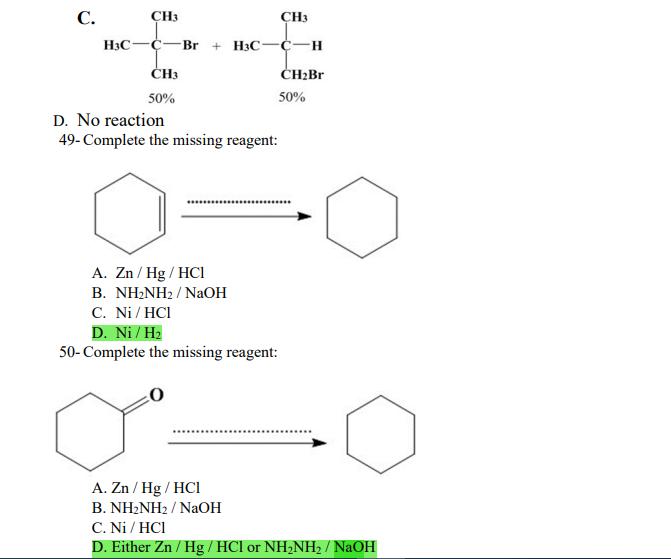
1- Alkanes are characterized by the general molecular formula: A. CnH2n-2 B. CnH2n C. CnH2n+2 2-Cycloalkanes are characterized by the general molecular formula: A. CnH2n-2 B. CnH2n C. CnH2n+2 3-What is the IUPAC name of the following compound? CH3 CH3-CH₂-CH₂-C-CH3 A. 4,4-dimethylpentane B. 1-tert-butylpropane 4-The common name of the following group is CH3CH₂CH- I CH3 CH3 A. n-butyl B. sec-butyl 5-Which one of the following is 2,2,5-trimethylhexane? A. (CH3)2CHCH2C(CH3)3 B. (CH3)2CHCH₂CH₂C(CH3)3 6-The correct IUPAC name of the following is CH3 HỌC-CH-CH2-CH-CH2-CH-CH-CH3 I CH3 I H₂C-CH3 A. 2,4,7-trimethylnonane. B. 7-ethyl-2,4-dimethyloctane. 7-What is the IUPAC name of the following? C. 2,2-dimethylpentane D. 1,1,1-trimethylbutane D. CnH2n+4 D. CnH2n+4 C. isobutyl C. CH3CH2CH(CH3)C(CH3)3 D. (CH3)2CHCH2 CH2CH2C(CH3)3 D. tert-butyl C. 3,6,8-trimethylnonane. D. 2-ethyl-5,7-dimethyloctane. 7-What is the IUPAC name of the following? CH₂CH3 CH3CH₂CH₂CH₂CHCHCH3 T CH₂CH3 A. 5,6-diethylheptane B. 5-ethyl-6-methyloctane C. 2,3-diethylheptane D. 4-ethyl-3-methyloctane 8-What is the IUPAC name of the following? A. 1-ethyl-4.4-dimethylcyclopentane B. 1-ethyl-3,3-dimethylcyclopentane 9-All the carbons in cyclopentane are A. primary carbons. B. secondary carbons. 10-The correct name of the following compound is C. 3-ethyl-1,1-dimethylcyclopentane D. 4-ethyl-1,1-dimethylcyclopentane C. tertiary carbons. D. quaternary carbons. A. (1-methylpropyl)cyclohexane. B. (2-methylpropyl)cyclohexane. 11-The correct IUPAC name of the following compound is C. (2,2-dimethylethyl)cyclohexane. D. (2,2-dimethylpropyl)cyclohexane. A. (1-methylhexyl)cyclopentane. C. 2-cyclopentylheptane. D. 1-cyclopentyl-2-heptane. B. (1-pentylethyl)cyclopentane. 12-The boiling point of isobutane (-10.2°C) is lower than n-butane (-0.4°C) because isobutane has A. weaker intermolecular van der Waals forces. B. stronger intermolecular van der Waals forces. C. weaker dipole-dipole attractive forces. D. stronger dipole-dipole attractive forces. 13-Arrange the following isomeric alkanes in order of increasing boiling point. I. n-heptane II. 2,3-dimethylpentane III. 2,2,3-trimethylbutane A. I < II < III B. II < III 14-Carbon atoms 1, 2, and 3 in the following structure are classified, respectively, as CH3 1 H3C 2 3 CH3 A. tertiary, primary, secondary. B. quaternary, secondary, secondary. C. quaternary, primary, tertiary. 15-Identify the isomer of C6H14 that only has primary and tertiary carbons. A. hexane B. 2,2-dimethylbutane 16-What is the total number of constitutional isomers with the formula C5H12? C. four A. two B. three 17-What is the IUPAC name of the following? A. 6-isopropyl-3-methylnonane B. 2-ethyl-5-isopropyloctane C. 3-methylpentane D. 2,3-dimethylbutane D. five C. 6-propyl-3-methylnonane D. 2-ethyl-5-propyloctane 18-The systematic name of the following group is H3C-CH-CH₂-CH₂-CH- I CH3 A. 5-ethyl-2-methylpentyl. B. 1-ethyl-4-methylpentyl. 19-What is the relationship between the two structures below? u and I H₂C-CH3 A. identical structures B. resonance forms C. constitutional isomers C. 6-methyl-3-heptyl. D. 2-methyl-5-heptyl. D. different compounds with different compositions 20-Arrange the following hydrocarbons in order of increasing boiling point. I. pentane II. 2,2-dimethylpropane III. 2-methylbutane A. I < II < III B. I < III < II 21-The 1,1-dimethylethyl group, -C(CH3)3, can also be called A. butyl. B. isobutyl. 22-What is the relationship between the following two structures? and C. II < I < III D. II < III 25-Which of the molecules below is NOT an isomer of formula C6H14? A H3C. A. A B. B 26-How many isomers of C4H9Cl are possible? H A. two B. three 27- Identify the conformation of butane shown below. CH3 H B A. anti H H C B. eclipsed x D C. C C. four C. skewed D. D D. five D. gauche 28- What is the IUPAC name of the compound shown in the following Newman projection? CH3 H H₂C H3C CH3 C. 1,2-dimethylethane D. 1,1,2,2-tetramethylethane 29- What is the dihedral (torsion) angle between the two bromine atoms in the following sawhorse drawing? Br CH3 H H A. 2,2,3,3-tetramethylbutane B. 2,3-dimethylbutane Br H CH3 A. 600 B. 00 C. 90⁰ D. 30⁰ 30- What is the IUPAC name of the compound shown below? CH3 H3C CH(CH3)2 H H A. 2,3,4-trimethylpentane B. 2-isopropylbutane 31- What is the IUPAC name of the following compound? H C. 1,2,3-trimethylbutane D. 2,3-dimethylpentane A. bicyclo[4.2.1]hexane B. bicyclo[6.3.0]heptane A. bicyclo[3.3.3]octane B. bicyclo[2.2.2]hexane 32- What is the IUPAC name of the following bicycloalkane? C. bicyclo[3.3.3]hexane D. bicyclo[2.2.2 Joctane C. bicyclo[4.1.0]hexane D. bicyclo[4.1.0]heptane 33- Identify the relationship between the following two Newman projections. H H₂C H H CH3 H H CH3 H CH₂ H₂C H and A. constitutional isomers B. stereoisomers 34- Which one of the following is the butane conformation shown below? H₂C H₂C A. anti B. eclipsed C. skewed 35- Which of the staggered conformations of 2-methylbutane is most stable? CH₂ H CH; CH₂ A. secondary carbons. B. quaternary carbons. CH3 CH₂ In Hi H CH₂ H A B 36- Cyclohexane is composed of A. methyl groups. B. methylene groups. 37- All the carbons in cyclopentane are H C. identical D. different conformations of the same compound HCH: D CH₂ H D. gauche C. methine groups. D. both methine and methylene groups. C. primary carbons. D. tertiary carbons. 38- What is the IUPAC name of the following structure? A. 3-ethylhexane B. 2-ethylheptane C. 4-ethylpentane D. 3-propylpentane 39- The sp3 orbitals of carbon in CH4 are formed from the A. three 2p orbitals. B. 2s and one of the 2p orbitals. C. 2s and two of the 2p orbitals. D. 2s and the three 2p orbitals. 40- The geometry of sp3 hybrid orbitals can be described as pointing towards the corners of a A. square. C. triangle. B. tetrahedron. D. square pyramid. 41-What is the Cl-C-Cl bond angle in CC14? A. 60 B. 90 C. 120 42- Which of the following statements is not true concerning hydrocarbons? A. Alkanes, alkenes, and alkynes are examples of aliphatic hydrocarbons. B. Aromatic hydrocarbons are also referred to as arenes. C. Hydrocarbons are compounds that carbon, hydrogen, and oxygen atoms. D. Hydrocarbons may contain sigma bonds and/or pi bonds. 43- The smallest straight-chain alkane that is liquid at room temperature and atmospheric pressure is A. butane. C. hexane. D. pentane. B. propane. 44- The lowest-boiling isomer of C7H16 would be XYY X C A B D 45- The hybridization of carbon atoms 1, 2, and 3 in the following are respectively, H,C=CH-CH3 1 2 A. sp, sp, and sp3. B. sp2, sp2, and sp3. 3 A. three 46- How many pi bonds are present in the following structure? H₂C=CH-CEN C. sp2, sp3, and sp³. D. sp, sp, and sp2. B. one D. 109.5 C. four D. two 47- The carbon-carbon single bond in the following is formed by the overlap of which two orbitals? H₂C=CH-C=N B. sp²-sp3 A. sp²-sp 48- Complete the following equation: CH3 A. B. H3C CH3 H3C -C-H CH₂Br CH3 CH3 H3C-C- Br CH3 H Br2/ hy C. sp2-sp2 D. sp-sp C. CH3 H3C C-Br+ H3C-C-H CH3 50% D. No reaction 49- Complete the missing reagent: CH3 CH₂Br 50% A. Zn/Hg/HCI B. NH,NHz / NaOH A. Zn/Hg/HCI B. NH2NH2/NaOH C. Ni/HCI D. Ni/H₂ 50- Complete the missing reagent: **** C. Ni/HCI D. Either Zn/Hg/HCl or NH₂NH₂/NaOH 1- Alkanes are characterized by the general molecular formula: A. CnH2n-2 B. CnH2n C. CnH2n+2 2-Cycloalkanes are characterized by the general molecular formula: A. CnH2n-2 B. CnH2n C. CnH2n+2 3-What is the IUPAC name of the following compound? CH3 CH3-CH₂-CH₂-C-CH3 A. 4,4-dimethylpentane B. 1-tert-butylpropane 4-The common name of the following group is CH3CH₂CH- I CH3 CH3 A. n-butyl B. sec-butyl 5-Which one of the following is 2,2,5-trimethylhexane? A. (CH3)2CHCH2C(CH3)3 B. (CH3)2CHCH₂CH₂C(CH3)3 6-The correct IUPAC name of the following is CH3 HỌC-CH-CH2-CH-CH2-CH-CH-CH3 I CH3 I H₂C-CH3 A. 2,4,7-trimethylnonane. B. 7-ethyl-2,4-dimethyloctane. 7-What is the IUPAC name of the following? C. 2,2-dimethylpentane D. 1,1,1-trimethylbutane D. CnH2n+4 D. CnH2n+4 C. isobutyl C. CH3CH2CH(CH3)C(CH3)3 D. (CH3)2CHCH2 CH2CH2C(CH3)3 D. tert-butyl C. 3,6,8-trimethylnonane. D. 2-ethyl-5,7-dimethyloctane. 7-What is the IUPAC name of the following? CH₂CH3 CH3CH₂CH₂CH₂CHCHCH3 T CH₂CH3 A. 5,6-diethylheptane B. 5-ethyl-6-methyloctane C. 2,3-diethylheptane D. 4-ethyl-3-methyloctane 8-What is the IUPAC name of the following? A. 1-ethyl-4.4-dimethylcyclopentane B. 1-ethyl-3,3-dimethylcyclopentane 9-All the carbons in cyclopentane are A. primary carbons. B. secondary carbons. 10-The correct name of the following compound is C. 3-ethyl-1,1-dimethylcyclopentane D. 4-ethyl-1,1-dimethylcyclopentane C. tertiary carbons. D. quaternary carbons. A. (1-methylpropyl)cyclohexane. B. (2-methylpropyl)cyclohexane. 11-The correct IUPAC name of the following compound is C. (2,2-dimethylethyl)cyclohexane. D. (2,2-dimethylpropyl)cyclohexane. A. (1-methylhexyl)cyclopentane. C. 2-cyclopentylheptane. D. 1-cyclopentyl-2-heptane. B. (1-pentylethyl)cyclopentane. 12-The boiling point of isobutane (-10.2°C) is lower than n-butane (-0.4°C) because isobutane has A. weaker intermolecular van der Waals forces. B. stronger intermolecular van der Waals forces. C. weaker dipole-dipole attractive forces. D. stronger dipole-dipole attractive forces. 13-Arrange the following isomeric alkanes in order of increasing boiling point. I. n-heptane II. 2,3-dimethylpentane III. 2,2,3-trimethylbutane A. I < II < III B. II < III 14-Carbon atoms 1, 2, and 3 in the following structure are classified, respectively, as CH3 1 H3C 2 3 CH3 A. tertiary, primary, secondary. B. quaternary, secondary, secondary. C. quaternary, primary, tertiary. 15-Identify the isomer of C6H14 that only has primary and tertiary carbons. A. hexane B. 2,2-dimethylbutane 16-What is the total number of constitutional isomers with the formula C5H12? C. four A. two B. three 17-What is the IUPAC name of the following? A. 6-isopropyl-3-methylnonane B. 2-ethyl-5-isopropyloctane C. 3-methylpentane D. 2,3-dimethylbutane D. five C. 6-propyl-3-methylnonane D. 2-ethyl-5-propyloctane 18-The systematic name of the following group is H3C-CH-CH₂-CH₂-CH- I CH3 A. 5-ethyl-2-methylpentyl. B. 1-ethyl-4-methylpentyl. 19-What is the relationship between the two structures below? u and I H₂C-CH3 A. identical structures B. resonance forms C. constitutional isomers C. 6-methyl-3-heptyl. D. 2-methyl-5-heptyl. D. different compounds with different compositions 20-Arrange the following hydrocarbons in order of increasing boiling point. I. pentane II. 2,2-dimethylpropane III. 2-methylbutane A. I < II < III B. I < III < II 21-The 1,1-dimethylethyl group, -C(CH3)3, can also be called A. butyl. B. isobutyl. 22-What is the relationship between the following two structures? and C. II < I < III D. II < III 25-Which of the molecules below is NOT an isomer of formula C6H14? A H3C. A. A B. B 26-How many isomers of C4H9Cl are possible? H A. two B. three 27- Identify the conformation of butane shown below. CH3 H B A. anti H H C B. eclipsed x D C. C C. four C. skewed D. D D. five D. gauche 28- What is the IUPAC name of the compound shown in the following Newman projection? CH3 H H₂C H3C CH3 C. 1,2-dimethylethane D. 1,1,2,2-tetramethylethane 29- What is the dihedral (torsion) angle between the two bromine atoms in the following sawhorse drawing? Br CH3 H H A. 2,2,3,3-tetramethylbutane B. 2,3-dimethylbutane Br H CH3 A. 600 B. 00 C. 90⁰ D. 30⁰ 30- What is the IUPAC name of the compound shown below? CH3 H3C CH(CH3)2 H H A. 2,3,4-trimethylpentane B. 2-isopropylbutane 31- What is the IUPAC name of the following compound? H C. 1,2,3-trimethylbutane D. 2,3-dimethylpentane A. bicyclo[4.2.1]hexane B. bicyclo[6.3.0]heptane A. bicyclo[3.3.3]octane B. bicyclo[2.2.2]hexane 32- What is the IUPAC name of the following bicycloalkane? C. bicyclo[3.3.3]hexane D. bicyclo[2.2.2 Joctane C. bicyclo[4.1.0]hexane D. bicyclo[4.1.0]heptane 33- Identify the relationship between the following two Newman projections. H H₂C H H CH3 H H CH3 H CH₂ H₂C H and A. constitutional isomers B. stereoisomers 34- Which one of the following is the butane conformation shown below? H₂C H₂C A. anti B. eclipsed C. skewed 35- Which of the staggered conformations of 2-methylbutane is most stable? CH₂ H CH; CH₂ A. secondary carbons. B. quaternary carbons. CH3 CH₂ In Hi H CH₂ H A B 36- Cyclohexane is composed of A. methyl groups. B. methylene groups. 37- All the carbons in cyclopentane are H C. identical D. different conformations of the same compound HCH: D CH₂ H D. gauche C. methine groups. D. both methine and methylene groups. C. primary carbons. D. tertiary carbons. 38- What is the IUPAC name of the following structure? A. 3-ethylhexane B. 2-ethylheptane C. 4-ethylpentane D. 3-propylpentane 39- The sp3 orbitals of carbon in CH4 are formed from the A. three 2p orbitals. B. 2s and one of the 2p orbitals. C. 2s and two of the 2p orbitals. D. 2s and the three 2p orbitals. 40- The geometry of sp3 hybrid orbitals can be described as pointing towards the corners of a A. square. C. triangle. B. tetrahedron. D. square pyramid. 41-What is the Cl-C-Cl bond angle in CC14? A. 60 B. 90 C. 120 42- Which of the following statements is not true concerning hydrocarbons? A. Alkanes, alkenes, and alkynes are examples of aliphatic hydrocarbons. B. Aromatic hydrocarbons are also referred to as arenes. C. Hydrocarbons are compounds that carbon, hydrogen, and oxygen atoms. D. Hydrocarbons may contain sigma bonds and/or pi bonds. 43- The smallest straight-chain alkane that is liquid at room temperature and atmospheric pressure is A. butane. C. hexane. D. pentane. B. propane. 44- The lowest-boiling isomer of C7H16 would be XYY X C A B D 45- The hybridization of carbon atoms 1, 2, and 3 in the following are respectively, H,C=CH-CH3 1 2 A. sp, sp, and sp3. B. sp2, sp2, and sp3. 3 A. three 46- How many pi bonds are present in the following structure? H₂C=CH-CEN C. sp2, sp3, and sp³. D. sp, sp, and sp2. B. one D. 109.5 C. four D. two 47- The carbon-carbon single bond in the following is formed by the overlap of which two orbitals? H₂C=CH-C=N B. sp²-sp3 A. sp²-sp 48- Complete the following equation: CH3 A. B. H3C CH3 H3C -C-H CH₂Br CH3 CH3 H3C-C- Br CH3 H Br2/ hy C. sp2-sp2 D. sp-sp C. CH3 H3C C-Br+ H3C-C-H CH3 50% D. No reaction 49- Complete the missing reagent: CH3 CH₂Br 50% A. Zn/Hg/HCI B. NH,NHz / NaOH A. Zn/Hg/HCI B. NH2NH2/NaOH C. Ni/HCI D. Ni/H₂ 50- Complete the missing reagent: **** C. Ni/HCI D. Either Zn/Hg/HCl or NH₂NH₂/NaOH
Expert Answer:

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
What is the IUPAC name of the compound shown? (a) (E )-3-Methyl-2-hexenoic acid (b) (Z )-3-Methyl-2-hexenoic acid (c) (E )-3-Methyl-3-hexenoic acid (d) (Z )-3-Methyl-3-hexenoic acid H;C CO.H C=C...
-
What is the IUPAC name of the compound shown in the margin? (a) Isopropyl 2-fluoro3-methylbutanoate; (b) 2-fluoroisobutanoyl 2-propanoate; (c) 1-methylethyl 2-fluorobutyrate; (d) 2-fluoroisopropyl...
-
What is the IUPAC name of this polymer? I-U-I I-C-H I-C-H I-U-H I-C-H I-C-H I-C-H I-U-H
-
This is one question with sub parts please solve this question step by step please also write the little explanation to solve the question Consider the following complex numbers: z, = 3+ 3i z2 = 3 +...
-
Select a chemical and identify the hazard associated. Discuss the proper sampling and analysis techniques according to NIOSH and OSHA. Research and discuss the methods and discuss whether there are...
-
State three different ways of expressing a periodic function in terms of its harmonics.
-
Briefly discuss the use of cash registers, prelists, and daily cash summaries in controlling cash receipts.
-
Robert Day has decided to run for a seat as Congressman from the House of Representatives, District 34, in Connecticut. He views his 8- month campaign for office as a major project and wishes to...
-
Evaluation a. Evaluate the effectiveness of social media marketing campaign for instagram, facebook and pintrest ?based on your KPIs for example account reached, content reached, likes, shares,...
-
The whistleblowing aspects of this case were first discussed in the text. What follows is a more comprehensive discussion of accounting and auditing issues. In 2005, Tony Menendez, a former Ernst &...
-
What is the companys Free Cash Flow (FCF)? Use the information to answer the following questions. XXX, Inc. Balance Sheet 2008 2007 2008 2007 Cash 450 200 Accounts Payable 1700 1500 Accounts...
-
Use Mason's gain rule to find transfer function of system shown in Fig. P3.10. R (s) 1 + 3 2 s +1 s + 1 6 5 7 Y (s)
-
For a particular product or service, such as Crest toothpaste or the Toyota Scion, how would you evaluate the countries that would represent the best prospects? Be specific. What information would...
-
Think of some brands that have high energy. What gives them that energy? Will that continue into the future?
-
Playtime Ltd produces jigsaws for sale in model shops. The following information relates to the sales and costs of producing the jigsaws. Selling price per unit is 20 Variable cost per unit is 10...
-
How is the idea of full costing extended when long-term environmental costs are considered?
-
Briefly describe some of the factors of labor productivity that are similar for some countries but different in others
-
A new car sold for $31,000. If the vehicle loses 15% of its value each year, how much will it be worth after 10 years?
-
The following additional information is available for the Dr. Ivan and Irene Incisor family. Ivan and Irene have the following investment income, in addition to that reported in Chapter 1: Dividends...
-
Bill and Guilda each own 50 percent of the stock of Radiata Corporation, an S corporation. Guilda's basis in her stock is $25,000. On July 31, 2012, Bill sells his stock, with a basis of $40,000, to...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-7. Ivan sold the following securities during the year and received a Form 1099-B that...
-
Discuss the approach you would take to building a system for playing Scrabble or another word game of the sort. What limitations does your system have? How likely do you think it is that your system...
-
Discuss the current state of the art of game-playing computer systems in relation to the following games: chess, checkers, Go, bridge, Othello, tic-tac-toe. What advances are likely in the near...
-
What problems did the developers of Chinook face? What new techniques did they add to simple Minimax with alpha-beta pruning? Would these techniques extend well to other games?

Study smarter with the SolutionInn App


