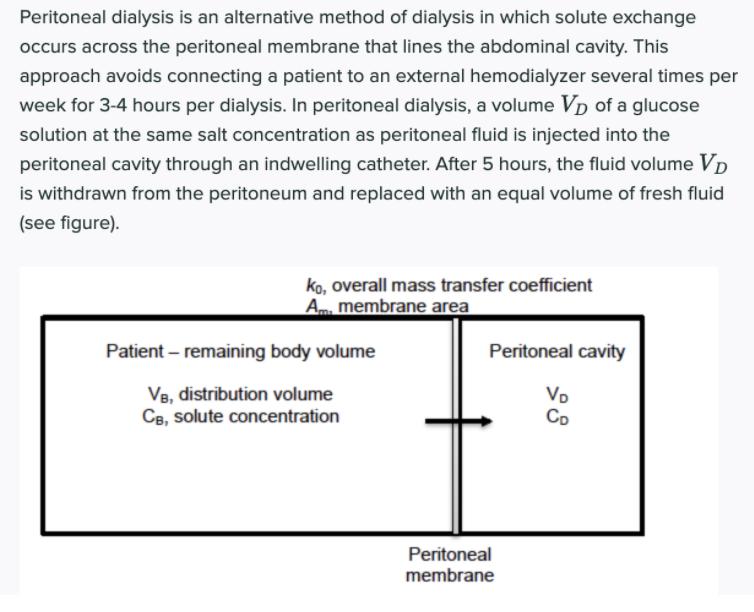
Peritoneal dialysis is an alternative method of dialysis in which solute exchange occurs across the peritoneal...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Peritoneal dialysis is an alternative method of dialysis in which solute exchange occurs across the peritoneal membrane that lines the abdominal cavity. This approach avoids connecting a patient to an external hemodialyzer several times per week for 3-4 hours per dialysis. In peritoneal dialysis, a volume Vp of a glucose solution at the same salt concentration as peritoneal fluid is injected into the peritoneal cavity through an indwelling catheter. After 5 hours, the fluid volume Vp is withdrawn from the peritoneum and replaced with an equal volume of fresh fluid (see figure). Ko, overall mass transfer coefficient Am membrane area Patient – remaining body volume Peritoneal cavity Vo Co V8, distribution volume CB, solute concentration Peritoneal membrane 1) Based on following data obtained for a 5-hr peritoneal dialysis session, determine the concentration of urea and vitamin B12 at the end of the exchange period as a fraction of the concentration in the body. Please comment based on a clinical point of view- specifically, what should a doctor suggest the dialysis patient do based on your answers? Data: Vp = 2 L; Va = 40 L; KAm = 21 cm' min (Urea) and K.Am = 5 cm' min" (Vitamin B12). Peritoneal dialysis is an alternative method of dialysis in which solute exchange occurs across the peritoneal membrane that lines the abdominal cavity. This approach avoids connecting a patient to an external hemodialyzer several times per week for 3-4 hours per dialysis. In peritoneal dialysis, a volume Vp of a glucose solution at the same salt concentration as peritoneal fluid is injected into the peritoneal cavity through an indwelling catheter. After 5 hours, the fluid volume Vp is withdrawn from the peritoneum and replaced with an equal volume of fresh fluid (see figure). Ko, overall mass transfer coefficient Am membrane area Patient – remaining body volume Peritoneal cavity Vo Co V8, distribution volume CB, solute concentration Peritoneal membrane 1) Based on following data obtained for a 5-hr peritoneal dialysis session, determine the concentration of urea and vitamin B12 at the end of the exchange period as a fraction of the concentration in the body. Please comment based on a clinical point of view- specifically, what should a doctor suggest the dialysis patient do based on your answers? Data: Vp = 2 L; Va = 40 L; KAm = 21 cm' min (Urea) and K.Am = 5 cm' min" (Vitamin B12).
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A chemist wishes to determine the concentration of CrO42- electrochemically. A cell is constructed consisting of a saturated calomel electrode (SCE; see Exercise 24) and a silver wire coated with...
-
A chemist wanted to determine the concentration of a solution of lactic acid, HC3H5O3. She found that the pH of the solution was 2.51. What was the concentration of the solution? The Ka of lactic...
-
A pharmaceutical manufacturer wants to determine the concentration of a key component of cough medicine that may be used without the drugs causing adverse side effects. As part of the analysis, a...
-
The total sales (all credit) of a firm are 6,40,000. It has a gross profit margin of 15 per cent and a current ratio of 2.5. The firm's current liabilities are 96,000; inventories 48,000 and cash...
-
A sample of manufactured CDs is obtained by using a computer to randomly generate a number between 1 and 1,000 for each CD, and the CD is selected if the generated number is 1,000. Identify which of...
-
Discuss the risk depicted by payroll system flowchart for problem 6. Describe the internal control improvements to the system that are needed to reduce theserisks. Supervisor Payroll Department...
-
In 2013, Thomas DePrince, a passenger aboard a cruise ship, visited the ships jewelry boutique, operated by Starboard Cruise Services, Inc. DePrince told the employees of the boutique that he was...
-
Reporting Net Sales and Expenses with Discounts, Returns, and Bad Debts The following data were selected from the records of Sykes Company for the year ended December 31, 2011.Balances January 1,...
-
(20%) A Fabry-Perot resonant cavity consists of a thin glass plate that has a refractive index of n = 1.50 and a thickness of = 100 m. Its surfaces are coated such that its peak transmittance is 100%...
-
The towline exerts a force of P = 4 kN at the end of the 20-m-long crane boom. If x = 25 m, determine the position ? of the boom so that this force creates a maximum moment about point O. What is...
-
Consider a monopolist with market inverse demand P(Q) = a-Q. The firm's production function is Q = F(L, K) = (L + K). The total cost of inputs is thus wL + rK. To maximize its profit, the firm must...
-
Q. Artisan Plastic and Processing (APP) wants to expand its operations. It is considering the purchase of pressing machine for its new line of plastic materials worth $800,000. The company estimates...
-
You just won a lottery that will pay you $500,000 a year for 10 years. If you deposit each of these payments in an account earning 8%, what will you have at the end of 10 years?
-
Which of these shows the result of using the first equation to substitute for y in the second equation, then combining like terms? y=3x 3x+2y=18
-
You are planning to make monthly deposits of $160 into a retirement account that pays 9 percent interest compounded monthly. If your first deposit will be made one month from now, how large will your...
-
Consider the information below about the following three stocks: Amethyst, Sapphire and Tanzanite. Market Price Amethyst $26 Sapphire $16 Tanzanite $10 Number of Shares 4 20 3 Cash $10 $65 $22 Debt...
-
1) Create a MongoDB database called "Seneca" 2) Create a single document in a collection called "student" with the following information. Note: the date format is month/day/year: student F name =...
-
3.16. For a system with non-identical service rates (see Sect. 3.5) and a limit of N jobs in the system (Eq. 3.13), obtain an expression for the mean service time per job, E[Ts], as a function of the...
-
In the presence of excess thiocyanate ion, SCN, the following reaction is first order in iron(III) ion, Fe3+; the rate constant is 1.27/s. Fe3+(aq) + SCN(aq) Fe(SCN)2+(aq) If 90.0% reaction is...
-
Write the Lewis formula that best describes the phosphoric acid molecule, H3PO4.
-
Sodium amide, known commercially as sodamide, is used in preparing indigo, the dye used to color blue jeans. It is an ionic compound with the formula NaNH2. What is the electrondot formula of the...
-
Mega Tech, Inc. designs and manufactures automotive components. For years, the company enjoyed a stable marketplace, a small but loyal group of customers, and a relatively predictable environment....
-
Describe the features of a project. How do they differ from day-to-day processes within an organization?
-
In 2003, the Department of Health and Human Services in Victoria, Australia, initiated a AU$323 million project to develop HealthSMART, an integrated IT system that would deliver resource management,...

Study smarter with the SolutionInn App


